A preview of this full-text is provided by American Society for Microbiology.
Content available from Journal of Clinical Microbiology
This content is subject to copyright. Terms and conditions apply.
JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 2010, p. 3008–3011 Vol. 48, No. 8
0095-1137/10/$12.00 doi:10.1128/JCM.02368-09
Copyright © 2010, American Society for Microbiology. All Rights Reserved.
CASE REPORTS
Subcutaneous Mycoleptodiscus indicus Infection in an
Immunosuppressed Dog
䌤
Catherine A. Metry,
1
Patricia S. Hoien-Dalen,
2
Carol W. Maddox,
3
Elizabeth H. Thompson,
4
Deanna A. Sutton,
4
Anna M. Romanelli,
5
Brian L. Wickes,
5
and Amy L. MacNeill
3
*
Department of Veterinary Clinical Medicine,
1
Veterinary Diagnostic Laboratory,
2
and Department of Pathobiology,
3
University of
Illinois, Urbana, Illinois, and Fungus Testing Laboratory, Department of Pathology,
4
and Department of Microbiology,
5
University of Texas Health Science Center, San Antonio, Texas
Received 3 December 2009/Returned for modification 20 January 2010/Accepted 21 May 2010
An 8-year-old dog presented with several dermal excoriations. Lesion cytology revealed pyogranulomatous
inflammation with branching, septate hyphae. A mold identified as Mycoleptodiscus indicus by morphology and
sequencing was cultured from fine-needle aspirates. This is the first report of a Mycoleptodiscus species as an
etiologic agent in a dog.
CASE REPORT
An 8-year-old, outdoor, male, castrated pointer dog was
presented to the University of Illinois Veterinary Teaching
Hospital in April 2009 for recheck blood work 2 months after
diagnosis of immune-mediated hemolytic anemia. Immuno-
suppressive treatment included daily oral administration of 2.1
mg prednisone/kg of body weight and 10.4 mg/kg cyclosporine
(Atopica; Novartis Animal Health), along with 20 mg aspirin to
prevent clot formation and 20 mg famotidine to limit gastro-
intestinal upset associated with the steroid administration. In
addition, 200 mg doxycycline had been given twice daily for
several weeks to treat for a possible underlying tick-borne
illness.
The general physical exam revealed a potbellied appearance,
hepatomegaly, and moderate to marked cachexia. The left rear
leg was swollen, with pitting edema and a draining tract on the
lateral aspect of the hock, the left popliteal lymph node was
markedly enlarged, and several areas of minor dermal excori-
ations were present along the nasal planum. In addition, the
dog’s weight had decreased from 38.5 kg to 35 kg over the
2-month time period. Clinical differentials for the draining
tract included phaeohyphomycosis, zygomycosis, pythiosis, lag-
enidiosis, sporotrichosis, and atypical bacterial infections, such
as nocardiosis, actinomycosis, and mycobacteriosis.
Fine-needle aspirates were acquired from the left popliteal
lymph node and from lesions on the bridge of the nose and the
distal aspect of the left hind leg. Aspirates were submitted for
cytologic evaluation, aerobic bacterial cultures, and fungal cul-
tures. All cytology samples were Wright-Giemsa stained and
were highly cellular, with a mixed population of severely de-
generate neutrophils and reactive macrophages, including ep-
ithelioid macrophages and multinucleated giant cells. Bacterial
cocci were noted extracellularly and within neutrophils. Aspi-
rates from all sites contained basophilic, septate hyphae which
measured 30 to 100 m in length and 3 to 6 m in width (Fig.
1). Some hyphae terminated in bulbous ends. The sample from
the popliteal lymph node also contained several lymphocytes
and rare, large, round yeastlike structures which rarely exhib-
ited a narrow-base bud. The samples were interpreted as
marked pyogranulomatous inflammation with bacterial and
fungal sepsis.
Pending results of bacterial and fungal cultures, itraconazole
was started at 5 mg/kg once daily, in addition to 32 mg/kg
terbinafine once daily and 29 mg/kg cephalexin twice daily. The
aerobic bacterial culture revealed a Staphylococcus pseudinter-
medius strain that was resistant to cephalexin but susceptible to
clindamycin, amoxicillin-clavulanate, and enrofloxacin. Cepha-
lexin was discontinued and clindamycin (12 mg/kg) once daily
was started.
Fungal cultures at the University of Illinois Veterinary Di-
agnostic Laboratory recovered a rapidly growing mold on Sa-
bouraud dextrose agar after 3 days of incubation at 25°C. The
colonies had a uniform morphological appearance, consistent
with isolation of a single species of fungus. By day 10, the
colonies had a dark-brown reverse with a white woolly surface.
Hyaline conidia were present at 2 weeks but were not readily
identifiable, so internal transcribed spacer (ITS) sequencing
was performed as described by Katsu et al. (6). A specific
sequence match was not available in the NCBI GenBank da-
tabase using the BLASTn algorithm in April 2009; therefore,
the isolate was submitted to the Fungus Testing Laboratory at
the University of Texas Health Science Center for additional
sequencing and morphological studies under the culture col-
lection number R-4334.
For morphological studies, the isolate was subcultured onto
potato flake agar, prepared in-house, and incubated at 25°C.
After 2 weeks, colonies were moderately fast growing, funicu-
lous, and gray-yellow with patches of white and exhibited a
* Corresponding author. Mailing address: University of Illinois, De-
partment of Pathobiology, 2001 S. Lincoln Ave., Urbana, IL 61802.
Phone: (217) 244-3950. Fax: (217) 244-7421. E-mail: almac@illinois
.edu.
䌤
Published ahead of print on 2 June 2010.
3008
yellow reverse and a diffusing yellow pigment. After 21 days,
colonies were dark gray and the yellow pigment was no longer
present. The isolate grew well at both 25°C and 35°C. Micro-
scopically (on slide culture preparations), abundant appresso-
ria and small aggregates of dark-brown cells (sporodochial
conidiomata) were present. Appressoria were brown, smooth,
not deeply lobed, and 0- or 1-septate (Fig. 2). Sporodochial
aggregates were irregular in size and shape (Fig. 3A). Conid-
iogenous cells were broadly ovate or ampulliform with a prom-
inent aperture and flared collarette (Fig. 3A and B). Conidia
were hyaline, smooth, thin walled, broadly lunate, and mostly
0-septate (rarely 1-septate) (Fig. 3C) and measured 5 to 6 m
at the widest part and 16 mto20m long. The presence and
number of polar and/or lateral appendages varied, ranging
from none to polar at only one or both ends to polar and lateral
on one or both sides (Fig. 3D). Appendage lengths varied and
were up to 12 m long. As the culture aged, conidial pro-
duction began to decrease, with virtually no conidiogenesis
occurring after week 4. The optimal time for observing
conidia appeared to be within the first 1 to 2 weeks following
subculture. Based upon the morphological features de-
scribed above, the isolate was identified as a Mycoleptodiscus
species (1, 10, 11).
Antifungal susceptibility testing was performed according to
the method described in CLSI approved standard M38-A2 (3).
Results read at 24 and 48 h were as follows: amphotericin B
(0.125 and 0.25 g/ml, respectively), itraconazole (0.25 and 0.5
g/ml), voriconazole (0.25 and 0.5 g/ml), posaconazole
(⬍0.03 and 0.06 g/ml), and terbinafine (0.015 and 0.015 g/
ml). Although no defined breakpoints are available for Myco-
leptodiscus species, the isolate appears susceptible to all agents
tested based upon normally achievable serum concentrations
using standard dosing regimens.
Template DNA was prepared from a fresh subculture of
R-4334, which had been grown on potato dextrose agar (Difco,
Detroit, MI) for 20 h at 30°C. The sample was suspended in 50
l of PrepMan Ultra reagent (Applied Biosystems, Foster
City, CA) and processed according to the manufacturer’s in-
structions. DNA amplification by PCR was performed in a
total volume of 50 l using 3 l of the PrepMan supernatant as
a template, high-fidelity Pfx50 DNA polymerase (Invitrogen,
Carlsbad, CA), and primers ITS1 and ITS4, as previously de-
scribed (12). In addition, D1/D2 amplicons were obtained us-
ing primers NL1 and NL4 and PCR conditions described pre-
viously (7, 9). PCRs were run in a PTC-100 thermocycler (MJ
Research, Watertown, MA) using a three-step protocol, which
consisted of 30 cycles with an annealing temperature of 58°C
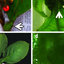
FIG. 1. Cytology of a fine-needle aspirate from a draining lesion on
the left hind limb of a dog showing branching, septate hyphae (filled
arrows). Bulbous structures at the terminal ends of the hyphae are also
depicted (open arrows). Wright-Giemsa stain. Magnification, ⫻500.
FIG. 2. Appressoria of this Mycoleptodiscus species as seen on a potato flake agar slide culture preparation mounted in lactophenol cotton
blue.
VOL. 48, 2010 CASE REPORTS 3009
and a 1-min extension time. The PCR products were cleaned
using a QIAquick PCR purification kit (Qiagen, Inc., Valencia,
CA). Purified templates were sequenced in both directions at
the University of Texas Health Science Center at San Antonio
Advanced Nucleic Acids Core Facility using the same primers
for ITS and D1/D2 amplification. Due to the paucity of se-
quence information available at the time of an initial search in
May 2009, we were unable to confirm the morphological iden-
tification. However, a subsequent ITS search of the NCBI
GenBank database in April 2010 revealed three hits at ⬎99.5%
identity (578/580, 576/577, and 567/568 nucleotides) at a ⬎90%
query coverage, corresponding to Mycoleptodiscus indicus
(GenBank accession no. GU980698, GU980694, and
GU980696). The next-closest species-level record was Myco-
leptodiscus terrestris, which showed only 79% identity at a 100%
query coverage. The D1/D2 BLASTn search showed similar
results. Two records were found with 100% identity (616/616
nucleotides) at a query coverage of 100%. These two isolates
were also M. indicus (accession no. GU980695 and
GU980693). A third M. indicus record (accession no.
GU980697) showed 99% identity but only at a 63% query
coverage.
As for the outcome of this case, the dog was seen at the
beginning of May 2009 and had multiple new subcutaneous
and dermal nodules along the rib cage and distal limbs.
Marked cellulitis was still present on the left distal rear limb.
Abdominal ultrasound was performed and revealed hepato-
megaly and a nonobstructive cholelith. Thoracic radiographs
indicated that the dog had generalized megaesophagus. Ra-
diographs of the distal aspect of the left tibia revealed cellulitis
and a tibial malunion fracture with bony proliferation. The
malunion fracture was thought to be an old injury; however,
mild osteomyelitis could not be ruled out. Epsom salt soaks
and whirlpool bath treatment were initiated in an effort to
decrease the cellulitis, and mupirocin was started topically to
help treat surface infections. At a subsequent visit, a slight
increase in respiratory effort was noted and the dog’s sclera
had a mild jaundiced appearance, consistent with hemolytic
anemia. The dog presented several days later to the emergency
service for lethargy, regurgitation, and aspiration pneumonia
secondary to megaesophagus. Clindamycin was discontinued,
as it may have contributed to the development of generalized
megaesophagus, and amoxicillin-clavulanate and enrofloxacin
were prescribed to treat the aspiration pneumonia.
Due to the severe clinical signs of iatrogenic hyperadreno-
corticism, several attempts were made to decrease the dosage
of prednisone being administered to this patient, but with each
attempt, the packed-cell volume decreased steadily over sev-
eral days and there was evidence of hemolysis. As a result, the
patient was maintained on oral prednisone (40 mg every 12 h)
FIG. 3. Microscopic features of this Mycoleptodiscus species. (A) Small aggregate of sporodochial cells, a conidiogenous cell, and a conidium
being released. Slide culture preparation mounted in lacto-fuchsin. (B) Conidiogenous cells with collarettes, and single-celled conidia with very
short appendages. (C) Two-celled conidium. (D) Cellophane tape preparation demonstrating single-celled conidia with both polar and lateral
appendages.
3010 CASE REPORTS J. CLIN.MICROBIOL.
and cyclosporine (400 mg daily), along with itraconazole, ter-
binafine, amoxicillin-clavulanate, and enrofloxacin, as previ-
ously described. The subcutaneous lesions waxed and waned
during the course of treatment. The dog was discharged from
the hospital on hospice care at the end of May and passed away
at his home on 12 June 2009, approximately 2 months after
diagnosis of this subcutaneous mycosis. A necropsy was not
performed.
Anamorphic fungi in the genus Mycoleptodiscus are in the
sexual-perithecium-forming family Magnaporthaceae. They
are typically thought of as plant pathogens; however, M. indi-
cus has been reported as an etiologic agent in immunosup-
pressed humans (5, 8) and, more recently, in a healthy human
male (4). Infections are presumably acquired by traumatic
implantation of the fungus.
Species identification of the etiologic agent in this case
proved difficult. The most informative phenotypic features for
identification of M. indicus include the presence of appressoria
(Fig. 2) (also seen in a few other clinically significant molds,
such as Colletotrichum species), dark phialidic, conidiogenous
cells with prominent collarettes aggregated into sporodochial
masses (Fig. 3A and B), and the production of large (10 to 16
by5to7m), hyaline, typically single-celled, curved conidia
(Fig. 3A and B). The production of septate conidia seen in the
initial morphological workup suggested M. terrestris (10, 11)
rather than M. indicus. The production of a yellow diffusible
pigment on potato dextrose agar noticed in a subsequent ex-
amination in combination with the shape of the appressoria
and the variable nature of the conidia also suggested two
species with lateral appendages, namely, Mycoleptodiscus varia-
bilis and/or Mycoleptodiscus lateralis (1).
Subsequent to submission of this report, ITS and D1/D2
nucleotide data for three human strains of M. indicus were
deposited into GenBank. These three isolates showed the
highest percent identities to our isolate, with all identities for
both loci being ⬎99.5%. These new deposits are from the
University of Alberta Microfungus Collection and Herbarium.
Since they are culture collection isolates and displayed good
query coverage, there is high confidence in the sequence iden-
tification.
The identification of this isolate as M. indicus by sequence
identity expands the known morphological features of this
species, as conidia that are septate and/or exhibit lateral
appendages have not been previously described. In addition,
this is the first reported case of subcutaneous M. indicus
infection in a dog.
Nucleotide sequence accession numbers. The D1/D2 se-
quence was deposited into GenBank under accession no.
GU220383. The ITS sequence was deposited under accession
no. GU220382. Our isolate was deposited into the University
of Alberta Microfungus Collection and Herbarium under ac-
cession number UAMH 11157.
B.L.W. is supported by grant PR054228 from the U.S. Army Med-
ical Research and Materiel Command, Office of Congressionally Di-
rected Medical Research Programs. A.M.R. is supported by NIDCR
grant DE14318 (CO STAR).
REFERENCES
1. Alcorn, J. L. 1994. Appressoria in Mycoleptodiscus species. Aust. Syst. Bot.
7:591–603.
2. Reference deleted.
3. CLSI. 2009. Reference method for broth dilution antifungal susceptibility
testing of filamentous fungi. Approved standard M38-A2. Clinical and Lab-
oratory Standards Institute, Wayne, PA.
4. Dewar, C. L., and L. Sigler. 12 April 2010. Fungal arthritis of the knee caused
by Mycoleptodiscus indicus. Clin. Rheumatol. [Epub ahead of print.] doi:
10.1007/s10067-010-1448-9.
5. Garrison, A. P., G. W. Procop, V. Vincek, J. Moon, M. I. Morris, S. Dobecki-
Lewis, T. J. Cleary, D. Brust, and I. Rosa-Cunha. 2008. A case of subcuta-
neous Mycoleptodiscus indicus infection in a liver transplant recipient suc-
cessfully treated with antifungal therapy. Transpl. Infect. Dis. 10:218–220.
6. Katsu, M., S. Kidd, A. Ando, M. L. Moretti-Branchini, Y. Mikami, K.
Nishimura, and W. Meyer. 2004. The internal transcribed spacers and 5.8S
rRNA gene show extensive diversity among isolates of the Cryptococcus
neoformans species complex. FEMS Yeast Res. 4:377–388.
7. Kurtzman, C. P., and C. J. Robnett. 1997. Identification of clinically impor-
tant ascomycetous yeasts based on nucleotide divergence in the 5⬘end of the
large-subunit (26S) ribosomal DNA gene. J. Clin. Microbiol. 35:1216–1223.
8. Padhye, A. A., M. S. Davis, A. Reddick, M. F. Bell, E. D. Gearhart, and L. von
Moll. 1995. Mycoleptodiscus indicus: a new etiologic agent of phaeohypho-
mycosis. J. Clin. Microbiol. 33:2796–2797.
9. Peterson, S. W., and C. P. Kurtzman. 1990. Phylogenetic relationships
among species of the genus Issatchenkia Kudriavzev. Antonie Van Leeuwen-
hoek 58:235–240.
10. Shearer, J. F. 2007. Some observations concerning microsclerotia and spore
production of Mycoleptodiscus terrestris in culture. Mycologia 99:88–90.
11. Watanabe, T., J. de D. Moya, J. L. Gonza´lez, and A. Matsuda. 1997. Myco-
leptodiscus terrestris from black pepper roots in the Dominican Republic.
Mycoscience 38:91–94.
12. White, T. J., T. D. Bruns, S. B. Lee, and J. W. Taylor. 1990. Amplification
and sequencing of fungal ribosomal RNA genes for phylogenetics, p.
315–322. In N. Innis, D. Gelfand, J. Sninsky, and T. C. White (ed.),
PCR-protocols and applications: a laboratory manual. Academic Press,
New York, NY.
VOL. 48, 2010 CASE REPORTS 3011
Content uploaded by Carol Maddox
Author content
All content in this area was uploaded by Carol Maddox
Content may be subject to copyright.