Content uploaded by Eiji Tanaka
Author content
All content in this area was uploaded by Eiji Tanaka
Content may be subject to copyright.
www.landesbioscience.com Plant Signaling & Behavior 1
Plant Signaling & Behavior 5:4, 1-4; April 2010; © 2010 Landes Bioscience
ARTICLE ADDENDUM
ARTICLE ADDENDUM
This manuscript has been published online, prior to printing. Once the issue is complete and page numbers have been assigned, the citation will change accordingly.
Key words: apical dominance, endo-
phyte, epiphyte, histology, shoot develop-
ment, witches’ broom symptom
Submitted: 12/02/09
Accepted: 12/02/09
Previously published online:
www.landesbioscience.com/journals/psb/
article/10834
Correspondence to: Eiji Tanaka;
Email: tanakae@ishikawa-pu.ac.jp
%HHIRHYQXS8EREOE)7TIGM½GMRWMXYZMWY-
alization of the pathogenic endophytic fungus
Aciculosporium take, the cause of witches’ broom
MRFEQFSS%TTP)RZMVSR1MGVSFMSP
¯41-((3-
%)1
Aciculosporium take causes continuous
shoot growth but maintains normal
leaf-arrangement and branching patterns
in the host plant, which eventually result-
ing in witches’ broom disease of bamboo.
An in situ hybridization technique with
a species-specific oligonucleotide probe
was recently used to demonstrate that
endophytic mycelia of A. take is predom-
inantly distributed in the intercellular
spaces of the shoot apical meristem of the
host. Endophytic hyphae in meristematic
tissues, which may produce auxin, are
responsible for continuous primordium
initiation within the shoot apex. Here
I examine another bamboo witches’
broom causal fungus, Heteroepichloë
sasae. Both species are biotrophic and
belong to family Clavicipitaceae: how-
ever, H. sasae does not cause continuous
shoot growth. Histological study showed
that H. sasae mycelia were distributed
superficially, even on shoot apical mer-
istems. These observations suggest that
when their stromata develop, endophytic
A. take destroys shoot apical meristem
and epiphytic H. sasae chokes the shoot
apex of the host. Stromata formation
consequently causes lateral bud out-
growth because of release from apical
dominance. This process repeats and
eventually results in the witches’ broom
symptoms.
Causal Fungi of Bamboo Witches!
Broom
Aciculosporium take (Ascomycota;
Clavicipitaceae) is a causal agent of bam-
boo witches’ broom disease in East Asia.
Colonized shoots by A. take continue to
grow in an acropetal sequence with very
thin stems and little leaves, although nor-
mal bamboo shoots cease to grow when
three to five leaves develop (Fig. 1). The
elongating shoot closely resembles a stolon
or a vine but not super-elongation dis-
eases such as bakanae disease. When the
stroma is formed at the shoot apex, lateral
buds grow out. Leaf arrangement and
branching patterns are maintained even
in colonized shoots. As with Epichloë /
Neotyphodium endophytes, external fun-
gal materials other than stromata are not
observed on plant surfaces. The location
of endophytic mycelia is probably involved
in well-regulated symptoms. The author
recently demonstrated that the endophytic
mycelium of A. take was predominantly
distributed in the intercellular spaces of
shoot apical meristems of the host bamboo
plant.1 Endophytic hyphae were visualized
by the in situ hybridization technique with
a species-specific oligonucleotide probe.
Heteroepichloë sasae (Ascomycota;
Clavicipitaceae) also causes witches’
broom in small bamboo plants (e.g., Sasa
spp.) in Japan and China.2,3 As with A.
take, this fungus is included in the family
Clavicipitaceae: their phylogenetic rela-
tionships were examined.4 Unlike A. take,
H. sasae does not cause continuous shoot
elongation. Its stroma encloses undevel-
oped leaves of the host and does not pen-
etrate the leaf tissue.2 Its habitat before
stroma formation, its association with the
host, and the mechanisms of symptom
development have not been addressed
thus far. In this addendum, a compara-
tive study was performed between A. take
and H. sasae to observe the development of
bamboo witches’ broom symptoms.
Mechanisms of bamboo witches’ broom symptom development caused
by endophytic/epiphytic fungi
Eiji Tanaka
Department of Environmental Science a nd Engineering; Ishikawa Prefectural University; Nonoichi-cho, Ishikawa Japa n
2 Plant Signaling & Behavior Volume 5 Issue 4
the surface of shoot apical meristem (Fig.
2F). These observations strongly suggest
that H. sasae is entirely an epiphyte. The
mycelial distributions of H. sasae are simi-
lar to that of Myriogenosora species that is
clavicipitaceous fungi and causes dwarfing
of host.5,6 Vegetative mycelia of both spe-
cies may enlarge to develop stromata.
Mechanisms of Bamboo Witches!
Broom Symptom Development
The developmental mechanisms respon-
sible for continuous shoot elongation
by A. take were discussed previously.1 In
brief, colonized shoots may not be able
to produce a sufficient amount of endog-
enous free IA A to expand leaves and
stems, whereas endophytic A. take hyphae
within the apical meristem may continue
to produce exogenous free IA A for induc-
ing primordium initiation and maintain-
ing apical dominance.1,7 This histological
study suggests that A. take stroma forma-
tion destroys the shoot apical meristem, as
a result of which endophytic hyphae pass
through the epidermis to form stroma. On
the other hand, witches’ broom disease
caused by H. sasae may develop as follows.
Superficial mycelia of H. sasae thicken and
become stroma, although it is unclear how
nutrients are obtained from the host. The
stroma prevents the leaf blade from expand-
ing and eventually chokes the shoot. These
observations indicate that both biotrophic
fungi finally terminate the function of the
host shoot apex when they form stromata.
Consequently, lateral buds start growing
because of release from apical dominance.
Repetition of this sequence may result in
symptoms of witches’ broom disease.
Endophytic hyphae of A. take appear
to regulate shoot development. However,
other endophytic clavicipitaceous fungi
have an insignificant effect on shoot
development. For example, essentially
symptomless Neotyphodium endophytes
growing entirely in the intercellular spaces
also inhabit the shoot apical meristem of
the host.8 The study of these host-fungus
relationships may provide insight into the
shoot development process.
H. sasae enclosed undeveloped leaves and
filled in the space between leaves (Fig.
2B). No mycelium was observed within
leaf tissue, except for stomatal apertures
(Fig. 2D). It is noted that H. sasae does not
seem to intrude even from the stomata.
Longitudinal sections of the colonized
shoot without stromata were observed to
examine the association of fungi with shoot
apex (Fig. 2E and F). Endophytic mycelia
of A. take were observed in the intercellu-
lar spaces of shoot apical meristem tissue
(Fig. 2E), as shown previously.1 In con-
trast, mycelia of H. sasae were observed on
Distribution of Mycelia in Host
Plant
Cross sections of colonized shoots with
stromata were observed to examine the
distribution of mycelia (Fig. 2A and B).
Sections were stained as mentioned in
the legend for Figure 2. Stroma of A. take
was formed on the host stem and was
connected to the internal mycelia (Fig.
2A). Endophytic mycelia were observed
in intercellular spaces of the stem tissue,
and hyphae emerged between epidermal
cells to form stroma (Fig. 2C). Stroma of
Figure 1. Bamboo shoot (Phyllostachys bambusoides) colonized by Aciculosporium take. The colo-
RM^IHWLSSXGSRXMRYIWXSKVS[ERH½REPP]KIRIVEXIWWXVSQEEXXLIWLSSXETI\MXLEWWQEPPPIEZIW
JVSQXLIVIWTIGXMZIRSHI EPXLSYKLRSVQEPWLSSXWLEZIXLVIIXS½ZIPIEZIW&EVGQ
www.landesbioscience.com Plant Signaling & Behavior 3
References
1. Tanaka E. Specific i n situ visualization of the patho-
genic endophytic fungus Aciculosporium take, t he
cause of witches’ broom in bamboo. Appl Environ
Microbiol 2009 ; 75: 4829-34.
2. Tanak a E, Tanaka C, Gafur A, Tsuda M. Heteroepichloë,
gen. nov. (Clavicipitaceae ; Ascomycotina) on bamboo
plants in Ea st Asia. Myc oscienc e 2002; 43 :87-93.
3. C heng Y. Identification of Heteroepichloë species on
Brachystachyum densifl orum by morphology and phy-
togeny. Journal of Beijing Forestr y Universit y 2009 ;
31: 84 -9 0.
4. Tana ka E, Tanaka C. Phylogenetic study of clavicipi-
taceou s fungi using aceta ldehyde dehydrogenase gene
sequence s. Myco science 2008 ; 49:115-25.
5. Rykard D, Bacon C, Luttrel l E. Host relat ions of
Myriogenospora atramentosa and Balansia epichloë
(Clavicipitaceae). Phytopatholog y 1985; 75:950- 6.
6. W hite JF Jr, Glenn A. A study of two fungal epibi-
onts of gr asse s: structural features, host relation-
ships and classification in the genus Myriogenospora
(Clavicipitales). Am J B ot 1994; 81:216-23.
7. Tanaka E , Tanaka C, Ishihara A, Kuwa hara Y, Tsuda
M. Indole-3-ac etic acid biosynthesis in Aciculosporium
take, a causal agent of witche s’ broom of bamboo. J
Gen Pla nt Pathol 2003 ; 69:1-6.
Figure 2 .*SV½KYVIPIKIRHWIITEKI
4 Plant Signaling & Behavior Volume 5 Issue 4
9. Mowr y R, Winkler C. The coloration of acidic car-
bohydrates of bacteria and fungi in tissue sec tions
with special reference to c apsules of Cr yptococcus
neoformans, pneumococci and staphylo cocci. Am J
Pathol 1956; 32: 628-9.
8. Ch ristensen MJ, Bennett RJ, Ansari HA, Koga H,
Johnson RD, Bryan GT, et al. Epichloë endophytes
grow by intercalar y hyphal extension in elongating
grass leaves. Fung al Genet Biol 2008 ; 45: 84-93.
Figure 2 . Comparisons between bamboo shoot tissue colonized by Aciculosporium take and Heteroepichloë sasae. Both species cause bamboo witches’
FVSSQHMWIEWI7 EQTPIW[IVI½\IHIQFIHHIHMRTEVEJ½RERHWIGXMSRIHEWWLS[RTVIZMSYWP]1 All colonized bamboos were collected at Kanazawa-shi,
Ishikawa, Japan (36°30'25''N, 136°37'59''E). (A– D) Sections stained by Alcian blue (pH 2.5) and periodic acid Schif f reaction methods.9 The magenta
color indicates fungal materials. Perithecia with asci were stained blue-purple. (A and B) Several images were combined into one large image because
XLI½IPHSJZMI[SFX EMRIHF]EQMGVSWGSTI[EWWQEPPIVXLERXLIWEQTPIHMQIRWMSRW%'VSWWWIGXMSRSJA . take stroma with perithecia. Bar, 500 Mm.
(B) Cross section of H. sasae stroma with perithecia. Bar, 1 mm. (C) Interface between A. take stroma and the host plant tissue. Endophytic mycelia
were observed in the intercellular spaces of plant tissue. Bar, 100 Mm. (D) Inter face between H. sasae stroma and host plant tissue. Mycelia were ob-
WIVZIHWYTIV½GMEPP]SRXLITPERXWYVJEGII\GITXJSVWXSQEXE&EVMm. (E) Longitudinal section of an A. take-colonized shoot of P. bambusoides. The
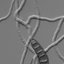
FPYIGSPSVMRHMGEXIWJYRKEPQ]GIPMEZMWYEPM^IHF]MRWMXYL]FVMHM^EXMSR[MXLEWTIGMIWWTIGM½GSPMKSRYGPISXMHITVSFI1 Endophytic hyphae were observed
in the intercellular spaces of shoot apical meristem (arrow) tissue. Bar, 100 Mm. (F ) Longitudinal section of an H. sasae-colonized shoot of Sasa palmata.
The green color indicates the fungal mycelia of H. sasaeWXEMRIH[MXL¾YSVIWGIMRMWSXLMSG]EREXIPEFIPIH[LIEXKIVQEKKPYXMRMR4PERXXMWWYI[EWWXEMRIH
[MXLWEJVERMRI1]GIPME[IVISFWIVZIHSRXLIWYVJEGISJXLIWLSSXETMGEPQIVMWXIQEVVS[8LIFVMKLX½IPHMQEKIERH¾YSVIWGIRXMQEKIW[IVIGSQ -
bined. B ar, 100 Mm.