Content uploaded by Noah Dunham
Author content
All content in this area was uploaded by Noah Dunham on Mar 07, 2023
Content may be subject to copyright.
Received: 2 August 2022
|
Revised: 16 January 2023
|
Accepted: 14 February 2023
DOI: 10.1002/zoo.21760
RESEARCH ARTICLE
Effects of honey locust seed pods on the behavior and
nutrient intake of zoo‐housed François langurs and
prehensile‐tailed porcupines
Noah T. Dunham
1,2
|Kaylin S. Tennant
1,2
|Andrew H. Loudon
3
|
Patricia M. Dennis
1,2,4
1
Division of Conservation and Science,
Cleveland Metroparks Zoo, Cleveland,
Ohio, USA
2
Department of Biology, Case Western
Reserve University, Cleveland, Ohio, USA
3
Department of Biology, Vancouver Island
University, Nanaimo, British Columbia, Canada
4
Department of Veterinary Preventive
Medicine, The Ohio State University,
Columbus, Ohio, USA
Correspondence
Noah T. Dunham, Cleveland Metroparks Zoo,
Cleveland, OH, USA.
Email: ntd@clevelandmetroparks.com
Funding information
Cleveland Metroparks Zoo
Abstract
Seed pods represent an under‐utilized and valuable dietary resource for zoos
because they encourage naturalistic extractive foraging behavior and because seeds
pods, like leafy browses, are more fiber‐rich than most dietary items typically offered
in zoos. The primary goal of this study was to examine the effects of honey locust
(Gleditsia triacanthos) seed pods on the behavior and macronutrient intake of zoo‐
housed François’langurs (Trachypithecus francoisi;n= 3) and prehensile‐tailed
porcupines (Coendou prehensilis;n= 2) using a pre‐versus postdiet implementation
design. From December 2019 to April 2020, we recorded behavior using
instantaneous interval sampling and daily macronutrient intake via dietary intake
records. We found that time spent feeding increased (p< .001) and stereotypic
behaviors decreased (p< .001) for the François’langur group during the seed pod
phase. The prehensile‐tailed porcupines also exhibited increased time spent feeding
and decreased inactivity (p< .001 for all comparisons) during the experimental seed
pod phase. We found no differences in macronutrient intake for the François’langur
group. The female prehensile‐tailed porcupine consumed more neutral detergent
fiber (NDF) in the seed pod phase (p= .003) and the male consumed more crude
protein, NDF, nonstructural carbohydrates, and crude fat (p< .001 for all compari-
sons). We stress that honey locust seed pods are a fiber‐rich (i.e., ~40%–55% NDF
by dry weight) dietary option for zoo‐housed folivores and promote positive welfare
by encouraging naturalistic foraging behavior and may help increase foraging time
and decrease stereotypic behaviors.
KEYWORDS
browse, Coendou prehensilis, diet, Trachypithecus francoisi
Zoo Biology. 2023;1–10. wileyonlinelibrary.com/journal/zoo
|
1
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium,
provided the original work is properly cited.
© 2023 The Authors. Zoo Biology published by Wiley Periodicals LLC.
10982361, 0, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/zoo.21760, Wiley Online Library on [07/03/2023]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
1|INTRODUCTION
For many zoo‐housed herbivores and folivores, leafy and woody
browses are used as enrichment or as a dietary supplement. Browse
offered at zoos typically consists of leaves and woody branches from
locally available trees, shrubs, and vines. Feeding browse can be
challenging for several reasons including, issues with seasonal
availability and storage, identifying browse species that are palatable
and nontoxic, discerning quantities of browse to offer, and quantify-
ing the consumption of various browse components (Clauss et al.,
2003; Clauss & Dierenfeld, 2008; Hatt & Clauss, 2006; Nijboer et al.,
2003). Despite these challenges, several studies have shown that the
addition of browse can be beneficial for animal health and welfare
(Cassella et al., 2012; Fuller et al., 2018; Lasky et al., 2021; Stoinski
et al., 2000).
Browse helps elicit natural foraging behaviors whereby animals
preferentially select particular browse parts (e.g., young leaves vs.
mature leaves; lamina vs. petiole) and may utilize unique manual and/
or oral processing behaviors to consume desired items. These
foraging behaviors, combined with the increased time required to
chew tougher and less calorically dense browse parts (Dunham &
Lambert, 2016), is often associated with increased time spent
foraging (Cassella et al., 2012; Fuller et al., 2018; Lasky et al.,
2021; Stoinski et al., 2000). The increase in time spent foraging
attributed to browse has also been associated with decreased
inactivity (Birke, 2002; Stoinski et al., 2000), decreased rates of
aggression (Fuller et al., 2018), and decreased rates of stereotypic or
undesirable behavior including pacing, head‐rolling, oral stereotypies,
and regurgitation and reingestion in a variety of taxa (Baxter &
Plowman, 2001; Cassella et al., 2012; Fuller et al., 2018; Koene &
Visser, 1996; Ruempler, 1992; Schmucker et al., 2010; Struck
et al., 2007).
In addition to its effects on behavior, browse can be beneficial
from a nutritional perspective. Browse generally has a nutrient profile
that better resembles items consumed by free‐ranging herbivores
and folivores. Most notably, browse typically contains greater fiber
concentrations and lower starch and sugar concentrations compared
to those of commercial biscuits, fruits, and vegetables commonly
offered in zoo settings (Dierenfeld et al., 2000,2021; Nijboer &
Dierenfeld, 1996; Remis & Dierenfeld 2004; Schmidt et al., 2005;
Smith et al., 2014; Wood et al., 2020). Elevated sugar and starch
levels from commercial diets are suspected to contribute to obesity
(Schwitzer & Kaumanns 2001), diabetes (Kuhar et al., 2013), and
dental issues (Cabana & Nekaris, 2015) in a variety of zoo‐housed
species. The higher fiber concentrations and plant secondary
metabolites found in browses have also been linked to improved
gastrointestinal health with foliage or browse‐heavy diets associated
with greater microbial diversity and lower relative abundances of
bacterial taxa associated with gastrointestinal inflammation (Amato
et al., 2016; Greene et al., 2018; Plocek et al., 2022).
Seed pods of the Fabaceae family represent a potentially under‐
utilized resource for zoo browse. Honey locust (Gleditsia triacanthos)
is a member of the Fabaceae family and is common throughout the
central and eastern United States (Shadow, 2008). These pods are
considered nontoxic and are regularly eaten by livestock and native
wildlife, including white‐tailed deer, squirrels, rabbits, opossums, and
raccoons (Nichols, 1958; Shadow, 2008). Like leafy and woody
browses, honey locust seed pods have higher fiber concentrations
relative to commercially available produce (Kamalak et al., 2012) and
may increase foraging time and encourage natural feeding behaviors,
including manual and/or oral processing behaviors required to extract
seeds (McGraw et al., 2016). Seed pods also have the potential to
freeze and store more effectively than leafy browse because seeds
are protected in outer casings.
We conducted preliminary feeding trials with honey locust
seed pods at Cleveland Metroparks Zoo in August 2019 and
identified that François’langurs (Trachypithecus francoisi)and
prehensile‐tailed porcupines (Coendou prehensilis)—two species
that consume Fabaceae seed pods in their native ranges (Charles‐
Dominique et al., 1981;Zhouetal.,2006,2009)—readily consumed
the pods. François’langurs are native to China and Vietnam and are
predominantly folivorous but also consume seed pods from the
Fabaceae family, particularly when young leaf availability is low
(Zhou et al., 2006,2009). Prehensile‐tailed porcupines are native to
the Amazon basin and are generally considered seed predators—
i.e., they rely heavily or almost exclusively on seeds from the
Fabaceae family for their daily nutrient intake during periods of the
year (Charles‐Dominiqueetal.,1981). The goals of the current
study were to first quantify the nutritional composition of honey
locust seeds pods throughout seasonal maturation and assess the
extent to which pods could be collected, frozen, and remain
palatable during winter months when leafy browse availability was
low. We then gradually implemented seed pods into the diets of
François’langurs and prehensile‐tailed porcupines and examined
the effects on behavior and macronutrient intake using a pre‐
versus postdiet implementation design. We predicted that, in both
study species, incorporating honey locust pods would significantly
increase time spent feeding, decrease inactivity and stereotypic
behaviors, and increase individuals’daily neutral detergent fiber
(NDF) intake.
2|MATERIALS AND METHODS
2.1 |Seed pod collection
Honey locust seeds pods were collected from trees on Cleveland
Metroparks Zoo grounds and nearby parks in July and August 2019.
Horticulture staff used a vehicle with an aerial work platform (i.e.,
“bucket truck”) to access seed pods located in tree canopies and used
shears to remove large clusters of seed pods. The authors then
trimmed off excess branching and discarded any open/damaged seed
pods. Seed pods required for the study were stored in two 55‐gallon
barrels and placed in a freezer. Approximately 3 h of staff time were
needed to collect and fill one 55‐gallon barrel with approximately
10 kg of seed pods.
2
|
DUNHAM ET AL.
10982361, 0, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/zoo.21760, Wiley Online Library on [07/03/2023]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
2.2 |Study subjects
We collected data on a group of François’langurs (n= 2 adult females
and 1 adult male) housed in an indoor habitat and two prehensile‐
tailed porcupines (n= 1 adult female and 1 adult male) housed in
separate indoor habitats at Cleveland Metroparks Zoo in Cleveland,
OH, USA.
2.3 |Diet change
This study included an 8‐week preseed pod baseline period (phase 1;
December 2019 to February 2020) followed by an 8‐week period
coinciding with the inclusion of honey locust seed pods in the diets of
François’langurs and prehensile‐tailed porcupines (phase 2; February
to April 2020). We monitored the behavior and macronutrient intake
throughout the entire 16‐week study duration. Seed pods were
gradually added to the diets for the first 2 weeks of phase 2 until they
constituted roughly 10%–20% of the diets by dry weight. Similarly,
seed pods were gradually reduced from the diets during the last 2
weeks of phase 2.
2.4 |Behavioral data collection
Behavioral data were collected on François’langurs and prehensile‐
tailed porcupines from Dec 2019 to April 2020 (n= 28 days for phase
1 and 28 days for phase 2). We used ANNKE 5MP Lite Security
Camera Systems (Rowland Heights) to record our study subjects in
their habitats and in their off‐exhibit holding areas. Due to issues with
one of the camera systems, only 25 days were recorded for the
female prehensile‐tailed porcupine during phase 2. Using video
footage, François’langur behavior was recorded using instantaneous
sampling at 10‐min intervals from 7:00 to 19:00. Given the similarity
in body size and general appearance of the three François’langurs, it
was not possible to identify individual langurs from the video footage.
Video footage was used to record prehensile‐tailed porcupine
behavior instantaneously at 15‐min intervals from 19:00 to 7:00
the following day (i.e., preliminary observations revealed the
prehensile‐tailed porcupines were active primarily at night and early
morning). Behaviors were recorded using ethograms unique to each
study species (Tables 1and 2).
2.5 |Macronutrient intake and macronutrient
analyses
We quantified food and macronutrient intake of our study subjects
throughout the study period. We first recorded the mass of each
food item offered per day. After controlling for moisture lost
overnight, we then subtracted the mass of each food item remaining
to quantify the mass of various food items consumed. Daily
macronutrient intake was quantified by multiplying the mass of each
food item consumed by the nutritional composition of each food
item. We used published nutritional composition values from Schmidt
et al. (2005) for the majority of food items (e.g., leafy greens, fruits,
and vegetables) offered to our study subjects. Food items not listed in
Schmidt et al. (2005), including commercial biscuits and honey locust
TABLE 1 Ethogram for François'
langurs. Behavior Description
Drink Ingesting water from food bowl or water tap
Feed Actively handling or chewing/ingesting food
Guest interaction Individual actively interacting with a guest through exhibit glass
Huddle Two or more individuals sitting in direct contact with one another. Arms or
legs of one individual are encompassing/embracing another
individual(s).
Inactive Stationary and not engaging in any obvious activity or other defined
behavior
Move Active locomotion including walking, running, climbing, and leaping
Object manipulate Interacting with enrichment item that does not involve feeding
Self‐direct Includes self‐grooming, scratching, self‐mouthing, and hair‐plucking
Social Includes both affiliative (e.g., initiating and receiving allogrooming, play,
and sexual behavior) and agonistic behaviors (baring teeth at another
individual and fighting)
Stereotypic Repetitive behaviors that lack obvious function including pacing, head‐
rolling, and swaying
Not visible Individual is out of site/out of view of recording cameras
Other Includes all behaviors not listed above
DUNHAM ET AL.
|
3
10982361, 0, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/zoo.21760, Wiley Online Library on [07/03/2023]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
seed pods, were shipped to Dairy One Forage Laboratory (Ithaca, NY,
USA) for macronutrient analyses following standardized procedures
for ash (AOAC Method 942.05), crude protein (CP: AOAC 990.03),
NDF (Van Soest et al., 1991), and crude fat (CF: AOAC 954.02). Total
nonstructural carbohydrate (TNC) was estimated via subtraction:
TNC = [100 −(% Ash + % CP + % NDF + % CF)]. Because the two
prehensile‐tailed porcupines were housed and fed separately, it was
possible to determine individual food and nutrient intake. We were
not able to differentiate food intake among members of the François’
langur group. Instead, we quantified total food and macronutrient
intake per day for the entire group and divided by the number of
individuals to generate an average food and macronutrient intake per
François’langur.
2.6 |Statistical analyses
G‐tests were used to compare overall activity budgets between
phases 1 and 2 for the study subjects. When activity budgets
differed significantly between phases 1 and 2, post hoc Z‐tests were
used to compare proportions of individual behaviors between
phases 1 and 2 (Gerstman, 2008). Multiple comparisons were
controlled for using Bonferroni corrections. The two prehensile‐
tailed porcupines were analyzed separately. Data for the François’
langur group were pooled and averaged given that individuals could
not be reliably identified from the camera footage. The male
François’langur died during the second week of phase 2 due to
cardiac disease and complications associated with his advanced age
(25 years), so the majority of the phase 2 dataset comes from two
individuals, while the phase 1 dataset is based on three individuals.
Rarely observed François’langur behavioral categories, including
drink, guest interaction, and object manipulation, were pooled into
the “other”category to facilitate statistical analyses. Due to
difficulties differentiating huddle versus inactive from the camera
footage, the two behaviors were pooled and reported as inactive.
Similarly, prehensile‐tailed porcupine behavioral categories were
condensed to include only feed, inactive, move, and other (i.e.,
“other”encompasses rarely observed behaviors including drink,
gnaw, object manipulation, and self‐direct). We did not observe any
stereotypic behaviors for either prehensile‐tailed porcupine. Non-
parametric Mann–Whitney U‐tests were used to compare macro-
nutrient intakes between phases 1 and 2 because intake data were
nonnormally distributed. We report daily macronutrient intake as
median values ± interquartile range.
2.7 |Ethical approval
The research protocol reported in this manuscript was approved by
Cleveland Metroparks Zoo's Scientific Research Committee and
Animal Care and Use Committee (protocol #CS2019‐028).
3|RESULTS
3.1 |Honey locust seed pod nutrient composition
and amounts consumed
We quantified the macronutrient content of honey locust seed pods
throughout different stages of maturation (Table 3). We primarily
collected seed pods at the unripe (i.e., green with seeds formed) stage
as preliminary tests indicated these were the most palatable for the
two study species. The François’langurs typically extracted and
consumed only the seeds while prehensile‐tailed porcupines typically
consumed the entire seed pod. Given this discrepancy, we used the
macronutrient content of “seeds only”versus “whole pods”when
quantifying the macronutrient intake of François’langurs and
prehensile‐tailed porcupines, respectively.
TABLE 2 Ethogram for prehensile‐
tailed porcupines.
Behavior Description
Drink Ingesting water from food bowl or water tap
Feed Actively handling or chewing/ingesting food
Gnaw Chewing on bark, caging, or other materials. Differs from feeding in that
objects are not consumed.
Inactive Stationary and not engaging in any obvious activity or other defined
behavior
Move Active locomotion including walking, running, climbing, and leaping
Object manipulate Interacting with enrichment item that does not involve feeding
Self‐direct Includes self‐grooming, scratching, self‐mouthing, and hair‐plucking
Stereotypic Repetitive behaviors that lack obvious function including pacing, head‐
rolling, and swaying
Not visible Individual is out of site/out of view of recording cameras
Other Includes all behaviors not listed above
4
|
DUNHAM ET AL.
10982361, 0, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/zoo.21760, Wiley Online Library on [07/03/2023]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
The François’langurs processed an average of 87.1 g of seed
pods per individual per day resulting in an estimate of 26.1 g of seeds
consumed per individual per day during phase 2 (range = 0–72 g of
seeds). Throughout the study, the François’langurs processed an
average of 49.1% of seed pods offered to them each day
(range = 0%–100%). The female prehensile‐tailed porcupine con-
sumed a mean of 22.3 g of seed pods per day during phase 2
(range = 0–112 g) or an average of 28.1% of seeds pods offered per
day. The male prehensile‐tailed porcupine consumed 40.6 g of seed
pods per day during phase 2 (range = 0–90 g) or an average of 45.4%
of seed pods consumed per day (Figure 1).
3.2 |Behavior
We found that the François’langur activity differed significantly
between phases 1 and 2 (G= 105.3; df =6; p< .001; Figure 2a).
Post hoc pairwise comparisons revealed that time spent feeding
significantly increased from phase 1 (10.5%) to phase 2 (14.4%;
Z=4.67; p< .001) and stereotypic behaviors (i.e., head‐rolling)
significantly declined from phase 1 (3.1%) to phase 2 (0.3%;
Z=7.93; p< .001). Overall activity budgets also differed signifi-
cantly between phases 1 and 2 for the female prehensile‐tailed
porcupine (G=28.3; df =3; p< .001; Figure 2b)andthemale
prehensile‐tailed porcupine (G= 67.6; df =3; p< .001; Figure 2c).
For the female, time spent feeding increased significantly from
phase 1 (6.1%) to phase 2 (11.6%) and time spent inactive
decreased from phase 1 (82.3%) to phase 2 (74.4%) (Z=4.78;
p<.001andZ=4.63;p< .001, respectively). For the male, all four
behavioral categories examined differed significantly from phases
1 to 2: feeding increased from 5.5% to 12.2% (Z=5.95;p< .001),
inactivity decreased from 77.4% to 63.8% (Z=7.56; p< .001),
moving increased from 15.8% to 21.1% (Z=3.47; p< .001), and
other increased from 1.3% to 2.9% (Z= 2.87; p= .004).
TABLE 3 Nutrient content of honey locust (Gleditsia triacanthos) seed pods throughout different stages of maturation.
Maturation stage Plant part % Moisture % CP
a
% NDF
a
% TNC
a
%CF
a
Unripe pod (green; seeds not yet formed) Whole pod
b
60.5 11.9 38.3 42.9 1.2
Unripe pod (green; seeds formed) Whole pod
b
70.7 13.2 49.1 28.6 2.4
Unripe pod (green; seeds formed) Seeds only 76.8 24.8 54.9 10.4 3.2
Ripe pod (dark brown/black; seed formed) Whole pod
b
12.0 8.6 28.2 50.9 1.8
Abbreviations: CF, crude fat; CP, crude protein; NDF, neutral detergent fiber; TNC, total nonstructural carbohydrate.
a
Values listed as percentage of dry mass
b
Includes exocarp, pulp, and seeds.
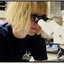
FIGURE 1 François’langur (left) and prehensile‐tailed porcupine (right) consuming honey locust seed pods. [Color figure can be viewed at
wileyonlinelibrary.com]
DUNHAM ET AL.
|
5
10982361, 0, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/zoo.21760, Wiley Online Library on [07/03/2023]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
3.3 |Macronutrient intake
The average daily macronutrient intake per François’langur did not differ
between phases 1 and 2 (Figure 3a).Thefemaleprehensile‐tailed
porcupine consumed significantly more NDF in phase 2 (18.7 ± 3.8 g)
compared to phase 1 (17.7 ± 2.7 g; U=9.1; df =1; p= .003; Figure 3b).
The male prehensile‐tailed porcupine consumed significantly greater
quantities of all macronutrients examined in phase 2 compared to phase 1
(Figure 3c), including CP (phase 1: 19.4 ± 5.0 g; phase 2: 22.4 ± 1.7 g;
U=52.3; df =1; p< .001), NDF (phase 1: 16.4 ± 3.3 g; phase 2:
24.0 ± 5.0 g; U=66.5; df =1; p< .001), TNC (phase 1: 50.9 ± 10.8 g;
phase 2: 57.6±3.8g; U= 50.1; df =1; p< .001), and CF (phase 1:
4.1 ± 0.8 g; phase 2: 4.5 ± 0.3 g; U= 50.3; df =1; p< .001).
4|DISCUSSION
Zoo animals, particularly herbivores and folivores, typically have
significantly reduced foraging/feeding times relative to their free‐
ranging conspecifics (Britt, 1998; Kerridge, 2005; Less et al., 2014;
Wiedenmayer, 1998). As a strategy to promote positive welfare, zoos
and other captive care facilities often develop feeding practices with
the aim of increasing time spent feeding while simultaneously aiming
to decrease inactivity and rates of undesirable behaviors (Cabana
et al., 2018; Chatpongcharoen et al., 2021; Dishman et al., 2009;
Fuller et al., 2018; Less et al., 2014). One example of such practice is
the provisioning of leafy or woody browse. In most facilities, browse
is given as part of an enrichment regime, though some institutions
now use browse as a staple diet item for various herbivorous species
(Campbell et al., 2001; Dunham et al., 2022; Hatt & Clauss, 2006;
Hatt et al., 2005; Nijboer & Dierenfeld, 1996; Nijboer et al., 2003).
We incorporated honey locust seed pods in the diets of François’
langurs and prehensile‐tailed porcupines and examined the effects on
behavior and macronutrient intake using a pre‐versus postdiet
implementation design. Overall, our behavioral predictions were
largely supported. For both study species, time spent feeding
increased significantly with the inclusion of seed pods. We also
found that the honey locust seed pods promoted naturalistic foraging
behaviors. The François’langurs extracted the seeds from the pod
casings, often rotating and reorienting the seed pods several times
FIGURE 2 Activity budgets during phases 1 and 2 for the François’langur group (a), female prehensile‐tailed porcupine (b) and male
prehensile‐tailed porcupine (c). Significant differences between phases 1 and 2 are denoted by an asterisk. [Color figure can be viewed at
wileyonlinelibrary.com]
6
|
DUNHAM ET AL.
10982361, 0, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/zoo.21760, Wiley Online Library on [07/03/2023]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
while using their hands and teeth to expose the seeds (Video S1).
During preliminary observations, we noted langurs took approxi-
mately 3–5 min to process one honey locust seed pod and
prehensile‐tailed porcupines took up to 8 min to consume one pod.
These handling and processing times translated to a roughly 40%
increase in time spent feeding for the François’langur group and
roughly 90% and 120% increases in time spent feeding for the female
and male prehensile‐tailed porcupines, respectively.
Although there was not a significant difference in François’
langur inactivity levels between phases 1 and 2, there was a
significant difference for both porcupines. We also observed a
decrease in the rate of François’langur stereotypic behavior after
adding the seed pods to the diet, as predicted. We suspect that the
seed pods played a role in the decreased observations of head‐rolling;
however, this result is also likely influenced by the death of the male
François’langur during the second week of phase 2. That is,
preliminary in‐person observations before this study revealed that
the male head‐rolled more frequently than the two females. Because
we were not able to reliably discriminate among individuals from the
camera footage, we were unable to discern the effects of the seed
pods versus the loss of the male on the observed rates of head‐
rolling. Though not clear from this study due to the loss of the male
langur, other work has shown that providing more naturalistic diets
and encouraging zoo animals to spend more time foraging is an
effective strategy to promote activity and potentially reduce
stereotypic and undesirable behaviors (Baxter & Plowman, 2001;
Cassella et al., 2012; Chatpongcharoen et al., 2021; Fuller et al.,
2018; Koene & Visser, 1996; Ruempler, 1992; Schmucker et al.,
2010; Struck et al., 2007).
We predicted that incorporating the seed pods into the diets of
our study species would lead to increased daily fiber intake.
Nutritional analyses revealed that the seed pods had high NDF
concentrations (i.e., relative to most commercially available fruits and
vegetables; see Schmidt et al., 2005) throughout their different
maturation stages, averaging approximately 40%–55% NDF by dry
weight for the unripe stages we fed to the François’langurs and
prehensile‐tailed porcupines. Despite reliably consuming the seeds
from the pods (i.e., ~ 50% offered during the study), François’langur
daily NDF intake did not increase from phases 1 to 2. In fact, daily
consumption of all quantified macronutrients was remarkably similar
from phases 1 to 2. This result is attributed to the François’langur
group reducing their intake of fiber‐rich commercial biscuits during
FIGURE 3 Daily macronutrient intake during phases 1 and 2 for the François’langur group (a), female prehensile‐tailed porcupine (b) and
male prehensile‐tailed porcupine (c). Significant differences between phases 1 and 2 are denoted by a bar and asterisk. CF, crude fat; CP, crude
protein; NDF, neutral detergent fiber, TNC, total nonstructural carbohydrates. [Color figure can be viewed at wileyonlinelibrary.com]
DUNHAM ET AL.
|
7
10982361, 0, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/zoo.21760, Wiley Online Library on [07/03/2023]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
phase 2 while consuming seeds from the pods. The female
prehensile‐tailed porcupine had significantly greater NDF intake with
the inclusion of the seed pods in the diet. The male prehensile‐tailed
porcupine had significant increases in all macronutrients quantified
because he consumed more of the seed pods offered compared to
the adult female and continued to eat the majority of his nonseed
pod diet items as well. While it was not possible to systematically
record body weights throughout the study, opportunistic voluntary
body weight measurements indicated that all of the study subjects
maintained relatively consistent body weights when comparing the
start to the end of the study: 7.1–7.2 kg and 6.6–6.4 kg for the two
female langurs, 6.2–6.1 kg for the female prehensile‐tailed porcupine,
and 5.5–5.8 kg for the male prehensile‐tailed porcupine (Figure S1).
Increasing dietary fiber can lead to improved satiety in zoo‐housed
animals. The higher fiber concentrations and plant secondary
metabolites found in leafy browses have also been linked to
gastrointestinal health with foliage or browse‐heavy diets associated
with greater microbial diversity and lower relative abundances of
bacterial taxa associated with gastrointestinal inflammation in some
primate species (Greene et al., 2018; Plocek et al., 2022). Future
research is required to assess the extent to which honey locust seeds
pods impact gut microbial composition and gastrointestinal health.
5|CONCLUSION
Our work suggests that honey locust seed pods can be included,
where available, alongside leafy browses as an important diet item
and an option for feeding enrichment for François’langurs and
prehensile‐tailed porcupines. We found that incorporating honey
locust seeds pods into the diets of François’langurs and prehensile‐
tailed porcupines increased time spent feeding significantly and
encouraged naturalistic extractive foraging behavior in both study
species. Like leafy browses, honey locust seeds pods provide a fiber‐
rich option that better resembles the nutritional composition of food
items consumed by free‐ranging folivores and seed predators. Honey
locust seed pods are easy to collect either by hand from low‐hanging
branches or by using a “bucket truck”to access seed pods higher in
the tree canopy. We found that approximately 3 h of staff time were
required to fill one 55‐gallon drum with approximately 10 kg of seed
pods, but the rate of collection depends on seed pod crop size and
accessibility to a given honey locust tree. Both of our study species
readily consumed freshly picked seed pods and those that were
frozen/thawed, including pods that were frozen for more than
12 months. This makes seed pods particularly attractive for zoos in
more northern climates that struggle to provide leafy browse
throughout the winter months. While our study focuses on the
effects of honey locust seed pod consumption on François’langurs
and prehensile‐tailed porcupines, we have since found that a variety
of other zoo‐housed species readily consume the seed pods,
including giraffe, agouti, and Indian crested porcupine. Finally, honey
locust is considered nontoxic and safe for animals to consume.
However, other Fabaceae seeds pods, including those of black locust
(Robinia pseudoacacia), are toxic; therefore, it is imperative that staff
accurately identify seed pods before they are offered to animals.
ACKNOWLEDGMENTS
Funding for this project was provided by Cleveland Metroparks Zoo.
We thank members of the Cleveland Metroparks Zoo Animal
Programs and Animal Care staff (Tad Schoffner, Lynn Koscielny,
Scott Parish, Miranda Beran, Kim Jansen, Emily Dickinson, Bri Duff,
Amanda Dailey, Tiffany Mayo, Patty Potter, Joe Ropelewski, Regina
Gerald, and Kelly Morgan) for their support and assistance recording
dietary intake data. We also thank Jeff Deluca, Ryan Marhefka, and
Tim Krynak for assistance during seed pod collection. We thank the
editor and two anonymous reviewers for their comments that
improved this manuscript.
CONFLICT OF INTEREST STATEMENT
The authors declare no conflicts of interest.
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the
corresponding author upon reasonable request. The data that
support the findings of this study are available from figshare at the
following link: 10.6084/m9.figshare.22128446.v1.
REFERENCES
Amato, K. R., Metcalf, J. L., Song, S. J., Hale, V. L., Clayton, J.,
Ackermann, G., Humphrey, G., Niu, K., Cui, D., Zhao, H.,
Schrenzel, M. D., Tan, C. L., Knight, R., & Braun, J. (2016). Using
the gut microbiota as a novel tool for examining colobine primate GI
health. Global Ecology and Conservation,7, 225–237.
Baxter, E., & Plowman, A. B. (2001). The effect of increasing
dietary fibre on feeding, rumination, and oral stereotypies in
captive giraffes (Giraffa camelopardalis). Animal Welfare,10(3),
281–290.
Birke, L. (2002). Effects of browse, human visitors, and noise on the
behaviour of captive orangutans. Animal Welfare,11, 189–202.
Britt, A. (1998). Encouraging natural feeding behavior in captive‐bred
black and white ruffed lemurs (Varecia variegata variegata). Zoo
Biology,17(5), 379–392.
Cabana, F., Jasmi, R., & Maguire, R. (2018). Great ape nutrition: Low‐sugar
and high‐fibre diets can lead to increased natural behaviours,
decreased regurgitation and reingestion, and reversal of prediabetes.
International Zoo Yearbook,52(1), 48–61.
Cabana, F., & Nekaris, K. A. I. (2015). Diets high in fruits and low in gum
exudates promote the occurrence and development of dental
disease in pygmy slow loris (Nycticebus pygmaeus): High fruit and
low gum diets promote dental disease innycticebus. Zoo Biology,34,
547–553.
Campbell, J. L., Glenn, K. M., Grossi, B., & Eisemann, J. H. (2001). ). Use of
local North Carolina browse species to supplement the diet of a
captive colony of folivorous primates (Propithecus spp.). Zoo Biology,
20(6), 447–461.
Cassella, C. M., Mills, A., & Lukas, K. E. (2012). Prevalence of regurgitation
and reingestion in orangutans housed in North American zoos and
an examination of factors influencing its occurrence in a single group
of Bornean orangutans. Zoo Biology,31, 609–620.
Charles‐Dominique, P., Atramentowicz, M., Charles‐Dominique, M.,
Gerard, H., Hladik, A., Hladik, C. M., & Prévost, M. F. (1981).
Les mamiferes arboricoles nocturnes d'une foret guyanaise:
8
|
DUNHAM ET AL.
10982361, 0, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/zoo.21760, Wiley Online Library on [07/03/2023]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
Interrelations plantes‐animaux. Revue d'Ecologie‐LaTerre et laVie,35,
341–435.
Chatpongcharoen, P., Campera, M., Laithong, P., Gibson, N. L., &
Nekaris, K. A. I. (2021). Naturalising diet to reduce stereotypic
behaviours in slow lorises rescued from wildlife trade. Applied Animal
Behaviour Science,242, 105413.
Clauss, M., Kienzle, E., & Wiesner, H. (2003). Feeding browse to large zoo
herbivores: How much is “a lot”, how much is “sufficient”.Zoo Animal
Nutrition,2,17–25.
Clauss, M. A., & Dierenfeld, E. S. (2008). The nutrition of browsers. Zoo
and Wild Animal Medicine: Current Therapy,6, 444–454.
Dierenfeld, E. S., Bezjian, M., & Dabek, L. (2021). Nutrient composition of
locally available browses consumed by Matschie's tree kangaroo
(Dendrolagus matschiei) in six North American zoological facilities.
Journal of Agriculture and Ecology Research International,21(11),
10–17.
Dierenfeld, E. S., Wildman, R. E. C., & Romo, S. (2000). Feed intake, diet
utilization, and composition of browses consumed by the Sumatran
rhino (Dicerorhinus sumatrensis) in a North American zoo. Zoo Biology,
19, 169–180.
Dishman, D. L., Thomson, D. M., & Karnovsky, N. J. (2009). Does simple
feeding enrichment raise activity levels of captive ring‐tailed lemurs
(Lemur catta)? Applied Animal Behaviour Science,116(1), 88–95.
Dunham, N. T., Koester, D. C., Dierenfeld, E. S., Rimbach, R., & Pontzer, H.
(2022). Balancing the scales: Preliminary investigation of total
energy expenditure and daily metabolizable energy intake in
Matschie's tree kangaroo (Dendrolagus matschiei). PLoS One,17(6),
e0270570.
Dunham, N. T., & Lambert, A. L. (2016). The role of leaf toughness on
foraging efficiency in Angola black and white colobus monkeys
(Colobus angolensis palliatus): Dunham and Lambert. American Journal
of Physical Anthropology,161, 343–354.
Fuller, G., Murray, A., Thueme, M., McGuire, M., Vonk, J., & Allard, S.
(2018). Behavioral and hormonal responses to the availability of
forage material in western lowland gorillas (Gorilla gorilla gorilla). Zoo
Biology,37,23–34.
Gerstman, B. B. (2008). Basic biostatistics: Statistics for public health
practiceJones and Bartlett. Publishers.
Greene, L. K., McKenney, E. A., O'Connell, T. M., & Drea, C. M. (2018). The
critical role of dietary foliage in maintaining the gut microbiome and
metabolome of folivorous sifakas. Scientific Reports,8, 14482.
Hatt, J. M., & Clauss, M. (2006). Browse silage in zoo animal nutrition ‐
feeding enrichment of browsers during winter. In A. Fidgett, M.
Clauss, K. Eulenberger, J. Hatt, I. Hume, G. Janssens, & J. Nijboer,
(Eds.), Zoo Animal Nutrition (III, pp. 201–204). Filander Verlag.
Hatt, J. M., Schaub, D., Wanner, M., Wettstein, H.‐R., Flach, E. J., Tack, C.,
Hassig, M., Ortmann, S., Hummel, J., & Clauss, M. (2005). Energy and
fibre intake in a group of captive giraffe (Giraffa camelopardalis)
offered increasing amounts of browse. Journal of Veterinary Medicine
Series A,52(10), 485–490.
Kamalak, A., Guven, I., Kaplan, M., Boga, M., Atalay, A. I., & Ozkan, C. O.
(2012). Potential nutritive value of honey locust (Gleditsia tria-
canthos) pods from different growing sites for ruminants. Journal of
Agricultural Science and Technology,14, 115–126.
Kerridge, F. J. (2005). Environmental enrichment to address behavioral
differences between wild and captive black‐and‐white ruffed lemurs
(Varecia variegata). American Journal of Primatology,66(1), 71–84.
Koene, P., & Visser, E. K. (1996). Tongue playing behaviour in captive
giraffes. Zeitschrift fur Saugetierkunde,62(2), 106–111.
Kuhar, C. W., Fuller, G. A., & Dennis, P. M. (2013). A survey of diabetes
prevalence in zoo‐housed primates. Zoo Biology,32(1), 63–69.
Lasky, M., Campbell, J., Osborne, J. A., Ivory, E. L., Lasky, J., & Kendall, C. J.
(2021). Increasing browse and social complexity can improve zoo
elephant welfare. Zoo Biology,40,9–19.
Less, E. H., Bergl, R., Ball, R., Dennis, P. M., Kuhar, C. W., Lavin, S. R.,
Raghanti, M. A., Wensvoort, J., Willis, M. A., & Lukas, K. E. (2014).
Implementing a low‐starch biscuit‐free diet in zoo gorillas: The
impact on behavior. Zoo Biology,33,63–73.
McGraw, W. S., van Casteren, A., Kane, E., Geissler, E., Burrows, B., &
Daegling, D. J. (2016). Feeding and oral processing behaviors of two
colobine monkeys in Tai Forest, Ivory Coast. Journal of Human
Evolution,98,90–102.
Nichols, J. T. (1958). Food habits and behavior of the gray squirrel. Journal
of Mammalogy,39, 376–380.
Nijboer, J., Clauss, M., & Nobel, J. (2003). Browse silage: The future for
browsers in the wintertime? European Association of Zoos and
Aquariums Nutrition Special Issue,3,6–7.
Nijboer, J., & Dierenfeld, E. S. (1996). Comparison of diets fed to
southeast Asian colobines in North American and European zoos,
with emphasis on temperate browse composition. Zoo Biology,15,
499–507.
Plocek, M. R., Amato, K. R., & Dunham, N. T. (2022). Effects of dietary
leafy browse on the gut microbiome of zoo‐housed black and white
colobus monkeys (Colobus guereza Trachypithecus francoisi) and
François’langurs. The 44th Meeting of the American Society of
Primatologists. Denver, CO, USA.
Remis, M. J., & Dierenfeld, E. S. (2004). Digesta passage, digestibility, and
behavior in captive gorillas under two dietary regimens. International
Journal of Primatology,25, 825–845.
Ruempler, U. (1992). The Cologne Zoo diet for lowland gorillas (Gorilla
gorilla gorilla) to eliminate regurgitation and reingestion. International
Zoo Yearbook,31, 225–229.
Schmidt, D. A., Kerley, M. S., Porter, J. H., & Dempsey, J. L. (2005).
Structural and nonstructural carbohydrate, fat, and protein compo-
sition of commercially available, whole produce. Zoo Biology,24,
359–373.
Schmucker, S. S., Kolter, L., & Nogge, G. (2010). Analysis of the fine
structure of oral activities in captivity unravels control of food intake
in giraffe (Giraffa camelopardalis). Giraffa,4(1), 49–50.
Schwitzer, C., & Kaumanns, W. (2001). Body weights of ruffed lemurs
(Varecia variegata) in European zoos with reference to the problem
of obesity. Zoo Biology,20, 261–269.
Shadow, R. A. (2008). Plant guide: Honey locust. United States Department
of Agriculture. https://plants.usda.gov/factsheet/pdf/fs_gltr.pdf
Smith, B. K., Remis, M. J., & Dierenfeld, E. S. (2014). Nutrition of the
captive western lowland gorilla (Gorilla gorilla gorilla): A dietary
survey: Captive gorilla diet survey. Zoo Biology,33.
Stoinski, T. S., Daniel, E., & Maple, T. L. (2000). A preliminary study of the
behavioral effects of feeding enrichment on African elephants. Zoo
Biology,19(6), 485–493.
Struck, K., Videan, E. N., Fritz, J., & Murphy, J. (2007). Attempting to
reduce regurgitation and reingestion in a captive chimpanzee
through increased feeding opportunities: A case study. Lab Animal,
36(1), 35–38.
Van Soest, P. J., Robertson, J. B., & Lewis, B. A. (1991). Methods for
dietary fiber, neutral detergent fiber and non‐starch polysaccharides
in relation to animal nutrition. Journal of Dairy Science,74,
3583–3597.
Wiedenmayer, C. (1998). Food hiding and enrichment in captive Asian
elephants. Applied Animal Behaviour Science,56(1), 77–82.
Wood, J., Koutsos, E., Kendall, C. J., Minter, L. J., Tollefson, T. N., &
Heugten, K. A. (2020). Analyses of African elephant (Loxodonta
africana) diet with various browse and pellet inclusion levels. Zoo
Biology,39(1), 37–50.
Zhou, Q., Huang, Z., Wei, X., Wei, F., & Huang, C. (2009). Factors
influencing interannual and intersite variability in the diet of
Trachypithecus francoisi.International Journal of Primatology,30,
583–599.
DUNHAM ET AL.
|
9
10982361, 0, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/zoo.21760, Wiley Online Library on [07/03/2023]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License
Zhou, Q., Wei, F., Li, M., Huang, C., & Luo, B. (2006). Diet and food choice
of Trachypithecus francoisi in the Nonggang Nature Reserve, China.
International Journal of Primatology,27(5), 1441–1460.
SUPPORTING INFORMATION
Additional supporting information can be found online in the
Supporting Information section at the end of this article.
How to cite this article: Dunham, N. T., Tennant, K. S.,
Loudon, A. H., & Dennis, P. M. (2023). Effects of honey locust
seed pods on the behavior and nutrient intake of zoo‐housed
François langurs and prehensile‐tailed porcupines. Zoo
Biology,1–10. https://doi.org/10.1002/zoo.21760
10
|
DUNHAM ET AL.
10982361, 0, Downloaded from https://onlinelibrary.wiley.com/doi/10.1002/zoo.21760, Wiley Online Library on [07/03/2023]. See the Terms and Conditions (https://onlinelibrary.wiley.com/terms-and-conditions) on Wiley Online Library for rules of use; OA articles are governed by the applicable Creative Commons License