Content uploaded by Stig Pedersen
Author content
All content in this area was uploaded by Stig Pedersen on Nov 30, 2023
Content may be subject to copyright.
Loss of European silver eel passing a hydropower station
By M. I. Pedersen
1
, N. Jepsen
1
, K. Aarestrup
1
, A. Koed
1
, S. Pedersen
1
and F. Økland
2
1
DTU-Aqua, Section for Freshwater Fisheries Ecology, Silkeborg, Denmark;
2
Norwegian Institute for Nature Research, Trondheim,
Norway
Summary
The aim of this study was to assess escapement success of silver
eels, Anguilla anguilla (L.), in a lowland river while passing a
reservoir and a hydropower station. It was hypothesized that
passage success would be lowest at the hydropower station and
that survival and migration speed would be highest in the free-
flowing river section upstream the reservoir. Forty-five female
silver eels 56–86 cm in length were tagged with acoustic
transmitters and released in November 2006. Their migration
was monitored via automatic listening stations (ALS) in
various sections of the river, covering a total migration
distance of 64 km. Survival and progression rate of down-
stream migration was highest in the upstream river section and
significantly lower in the reservoir. The eels apparently had
trouble finding their way past the turbines and spent between
1.5 and 35 h in the forebay. The results show that within the
study period, only 23% of the tagged eels reached the tidal
limit, mainly due to difficulties in passing the hydropower dam.
With such high loss-rates, the escapement goals set in the
management plan cannot be achieved.
Introduction
The European eel population (Anguilla anguilla L.) has
declined considerably over the past decades. The present
recruitment of glass eels is only about 1–9% of the late-1970s
level, indicating a historically low spawning stock in the
Sargasso Sea (ICES, 2009). Consequently, the International
Council for the Exploitation of the Sea (ICES) stated that the
eel stock was beyond safe biological limits and recommended
that anthropogenic activities affecting the stock be reduced as
much as possible (ICES, 2001). This led to a proposed EU
recovery plan for the eel populations (EU, 2007). As part of
this plan, management measures must be implemented to meet
a target of 40% silver eel escapement from individual river
basins or eel management units.
Silver eel river migrations occur mainly during autumn, but
some migration activity is also observed in the spring
(Aarestrup et al., 2008). The timing of silver eel runs has been
related to the increases in water discharge (Haraldstad et al.,
1984; Vøllestad et al., 1986), lunar phases, and water temper-
ature (Vøllestad et al., 1986; Tesch, 2003). Migration activity
is, in general, nocturnal (Tesch, 2003).
To acquire knowledge on the behaviour and escapement of
migrating silver eels, a series of studies was conducted in the
River Gudenaa, Denmark. Previous studies investigated
behaviour and survival in the lower river and estuary (Aarest-
rup et al., 2008, 2010). The present study focuses on the middle
part of the river where downstream-migrating fish have to pass
free-flowing river sections, a reservoir and a hydropower
station.
The behaviour of downstream migrating eels at hydropower
stations has been the subject of a number of studies (i.e. Haro
et al., 2000; Behrmann-Godel and Eckmann, 2003; Durif
et al., 2003; Gosset et al., 2005; Boube
´e and Williams, 2006;
Calles et al., 2010). The general finding is that A. anguilla
approaching a power station often hesitate to continue their
migration, and only after repeated attempts may pass the
hydropower station either through the turbines, spillways or
the bypass passages. The two principal direct causes of
mortality at hydropower stations are turbine-related mortal-
ities and impingement on the screens (Monten, 1985; Calles
et al., 2010). Carr and Whoriskey (2008) found 100% mortal-
ity of American eels (Anguilla rostrata) descending through
turbines at a hydropower station. Winter et al. (2006) demon-
strated that fishing and hydropower were the main causes for
mortality of migrating eel in the River Meuse. Breteler et al.
(2007) found that only 23% of silver eels had successfully
migrated through the lower 300 km of the Rhine River.
The objective of this study was to assess overall escapement
success of silver eels migrating through regular river sections, a
reservoir and a hydropower station. For future eel manage-
ment it is imperative that knowledge on escapement is
available. It was hypothesized that passage success would be
lowest at the hydropower station and that survival and
migration speed would be highest in the free-flowing river
section upstream the reservoir.
Materials and methods
The River Gudenaa (55
o
52¢N, 9
o
33¢E) is a lowland stream
160 km long with a catchment area of 2631 km
2
and a mean
annual discharge of 32 m
3
s
)1
. The river system is strongly
influenced by human activities, including canalization, nutrient
loading from agriculture, hydropower damming, and develop-
ment. There are seven small hydropower stations on the River
Gudenaa. The largest and lowermost is the Tange Hydro-
power station (Fig. 1) with a catchment of 1699 km
2
(65% of
the total catchment) and a mean annual discharge of 21 m
3
s
)1
at the station. The station is situated 35 km from the tidal limit
and has three Francis turbines. The reservoir, Lake Tange, has
a surface area of 537 ha, length of 8.5 km and with a depth of
<8 m.
To protect migrating fish, Danish legislation requires
physical screens with a maximum bar distance of 10 mm to
be installed in front of hydroelectric turbines. This is also the
case at the Tange hydropower station, with 10 mm screens that
should keep silver eels from entering the turbines. There are
J. Appl. Ichthyol. 28 (2012), 189–193
2011 Blackwell Verlag, Berlin
ISSN 0175–8659
Received: April 4, 2011
Accepted: September 13, 2011
doi: 10.1111/j.1439-0426.2011.01913.x
U.S. Copyright Clearance Centre Code Statement: 0175–8659/2012/2802–0189$15.00/0
Applied Ichthyology
Journal of
two fish bypass facilities (Fig. 2), one is a Denil type fish ladder
with resting pools and with an entrance situated approximately
200 m upstream the turbines (Fig. 2). The other bypass system
consists of three (30 cm Ø) tube bypasses installed to help
downstream-migrating fish, especially salmonid smolts, to
bypass the turbines. One tube bypass is placed in each of the
three turbine screens approximately 0.5 m below the water
surface. Water discharge in the two different bypass systems
fluctuates with the number of turbines in use. The bypass flow
from each turbine in use is 150 L s
)1
. In the fish ladder there is
a constant flow of 150 L s
)1
. When all three turbines are in
use, the maximum discharge in the bypasses is 600 L s
)1
,
<3% of the mean total discharge. A light fence is installed to
guide eels to the fish ladder entrance (Fig. 1), but the effect of
this has never been evaluated.
In the study area there is one commercial fisherman
operating in the Tange reservoir using pound nets. Cormo-
rants Phalacrocorax carbo sinensis can be observed foraging in
the reservoir. The birds are possibly from a colony with 50–200
nests approximately 15 km from the reservoir.
The studied river stretch is 63.5 km. The study area was
divided into five sections (Fig. 1). The first section covers a
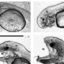
Fig. 1. Map of study area showing lower and middle part of River Gudenaa, Denmark. Automatic detection stations installed at all Sites 0–5.
River width varies from 15 to 30 m. Mean annual discharge at Site 3 was 21 m
3
s
)1
Fig. 2. Schematic diagram of Tange hydropower station with position of fish ladder and bypass in the deflecting screens in front of the three
Francis turbines
190 M. I. Pedersen et al.
distance of 4.1 km of free flowing river. The second section is
14.2 km of river and ends at the inlet of the reservoir. The third
section is the reservoir including the power canal (8.5 km). The
fourth section is the area from the end of the inlet canal to the
listening station 3.5 km downstream of the hydropower
station. The fifth section goes to the tidal limit and is
33.3 km long.
Forty-five downstream migrating silver eels were captured in
the autumn of 2006 in a permanent eel trap at the Vestbirk
hydropower station approximately 30 km upstream the release
site. Eels caught in the trap were kept in net-pens in the river
for 1–7 days before tagging. Eels were transferred to the
laboratory of DTU Aqua on 1–2 November and tagged by
surgical implanting with THELMA Ltd., Norway, LP-9
acoustic transmitters (9 ·34 mm, weight in air of 5.3 g,
weight in water of 3.3 g, guaranteed life time of 148 days)
using the method described by Aarestrup et al. (1999). The
tags had a programmed random interval range between
transmitted signals. Mean body length of the tagged fish was
66.3 ± 7.2 cm (SD) (range 56–86 cm), mean body mass
551 ± 213 g (range 303–1309 g). After tagging the eels were
kept in recovery tanks, from 1 to 8 h and by dusk released in a
small tributary approximately 300 m upstream the confluence
with the River Gudenaa. All tagged eels were released on 1–2
November.
Twelve hydrophone buoys (ALS, VR2; VEMCO Ltd.,
Canada) were placed pairwise at six sites (detection stations)
in the river (Fig. 1). These were continuously in operation
from 15 October 2006 to 25 March 2007. At each site, two
hydrophones were moored in the river approximately 25 m
apart. The ability of the VR2 to detect acoustic signals in a
range wider than the river was tested on all stations. In the
power-channel of the hydropower station significant acoustic
noise occurs as a consequence of the turbines. Four VR2s were
placed to cover the entire water column: two close to the
surface and two at the bottom. A single VR2 was placed to
detect upstream migration following release (Site 0, Fig. 1) and
one VR2 was placed at the tidal limit (Site 5).
Migration (progression) speed was calculated as the time
between first detection at site tand first detection at site t+1
divided by distance (d).
Statistical analysis
To test whether progression speed in the river section 1
between the detection station 1–2 could explain survival of the
eel to the tidal limit, a logistic regression with survived ⁄dead
as the dependent variable and progression speed in river
section 1 as the predictor variable was performed, and body
length and body weight were entered in the model as
covariates.
Progression speed was compared between two river sections
and the reservoir by repeated-measures
ANOVAANOVA
(r-m
ANOVAANOVA
)
with one within-subject factor (compartment). R-m
ANOVAANOVA
was
preferred to a profile analysis (
MANOVAMANOVA
) that has lower
statistical power (Maxwell and Delaney, 1990; Potvin et al.,
1990). To compensate for violation of the sphericity assump-
tion, the degrees of freedom were decreased by multiplication
by the Huynh-Feldt epsilon, which is a less conservative
adjustment than Greenhouse-Geisser and is recommended
when sample sizes are small (Von Ende, 1993; SPSS, 1997).
The data was log (X+ 1) transformed to meet the require-
ments of parametric analysis (i.e. normality and homoscedas-
ticity). Tests of within-subjects contrasts were achieved by a
repeated design and Bonferroni adjusted (Von Ende, 1993).
Results
Two of the 45 tagged eels were not detected after release at any
of the detection stations. Their fate is unknown and they are
therefore omitted from further analyses.
Forty-three eels resumed downstream migration after
release. Of these, 10 eels were detected at the tidal limit
(Fig. 1, Table 1). Thus, during the study period (1 November
2006–25 March 2007), the observed overall passage ⁄success of
tagged eels entering the first station to the estuary is 23%. Eels
were lost in the reservoir (n = 5) and in the last section of the
river (n = 6), but the major loss occurred between Sites 3 and
4 where the eels had to pass the Tange Hydropower station.
Here, 38 eels were detected in the inlet, but only 16 eels were
detected at the next downstream station during the study
period. Thus, 22 eels did not pass the power station and
subsequently the loss was considerably higher at the station
than at all other sections.
Following release, the tagged eels (n = 43) moved down-
stream after 0–38 days (mean 9.2 days). No tagged eels were
detected at the upstream detection station Site 0. Seventeen
eels quickly resumed migration and were detected at Site 1
within 24 h. One eel migrated on day two. Fourteen eels were
detected between 7 and 12 days post release, and between days
12 and 38 the remaining 11 eels were detected at Site 1. Once
having resumed migration, most eels (n = 40) moved quickly
downstream to the reservoir at a mean speed of 3.4 km h
)1
range (0.8–5.9 km h
)1
). The majority of eels reached the
reservoir within the same night as they resumed their migra-
tion. Three eels, however, spent 6, 10 and 14 days before they
reached the reservoir (Site 2).
After arriving in the reservoir the migrants spent an average
8.2 days (range 0.2–34.8 days) (n = 38) before entering the
power canal (Site 3). Most of the eels approached the station
several times over several days before entering, and seven of
the 38 eels detected in the canal reversed their direction of
migration and passed back and forth between the inlet and the
outlet of the reservoir (Sites 2 and 3), a distance of 8.5 km.
After arriving at the power canal the second time, three
individuals finally passed the hydropower station.
The eels clearly had problems locating ⁄entering the bypass
facilities, illustrated by the fact that they spent much more time
passing the detection station in the canal than they did at the
other stations (Table 1).
The 16 eels that succeeded in passing the hydropower station
spent from 2 h to 63 days with a mean of 11 days 5 h before
passing. The last 33.1 km to reach the tidal limit (Site 4–5)
were completed on average in 1.3 days (range 0.42–6.35 days).
The logistic regression showed no effect of the progression
rate in the river section between Sites 1 and 2, body length or
body weight on the loss of eels (P > 0.228).
The r-m
ANOVAANOVA
revealed that the progression speed differed
among the three sections Sites 1–2, 2–3 and 3–4 (P < 0.0001).
Progression speed was not statistically dependent on body
length or body weight (P > 0.857). None of the interaction
terms was statistically significant (P > 0.868). The Within-
Subjects contrast test showed that there was a significant
difference between sections Sites 1–2 and 2–3 (P < 0.0001),
and no significant difference between sections Sites 2–3 and
3–4, respectively (P = 0.132).
Loss of European silver eels 191
Discussion
Overall escapement to the tidal limit from the investigated
63.5 km river stretch was 23% in the autumn ⁄winter 2006. This
does not necessarily mean that the remaining 77% of the tagged
eels died. It is a well-known fact that some silver eels halt their
autumn migration and resume it the following spring or autumn
(Winter et al., 2007;Aarestrup et al., 2008;Simon and Fladung,
2009). Own (unpublished) results from PIT-tagged eels in the
Gudenaa show that approximately 10% resume migration the
following spring. However, the observed 58% loss of silver eels
during hydropower station passage is substantial, and in
combination with other causes of loss, constitutes a major
problem for the eel. Aarestrup et al. (2008, 2010) and found a
60–80% loss of A. anguilla moving through the Gudenaa
estuary, presumably caused by fishing. The combination of
hydropower station passage and estuarine fishing leaves very
little chance for silver eels from the upper or middle Gudenaa
catchment to reach the sea. The probability of reaching the
estuary was not related to initial progression speeds observed at
the first upriver section (Site 1–2). Progression speeds between
sections varied and were found to be significantly different
between river Site 1–2, the reservoir (Site 2–3), and the power
station (Site 3–4). The differences in progression speeds were not
related to size of the fish (length or weight); this is in contrast to
movement monitored through the estuary where the larger eels
did move faster (Aarestrup et al., 2010). Progression in the river
was relatively fast, but slow through the reservoir. The delay at
the reservoir and the hydropower station may increase the risk
of predation from fishermen and cormorants and are likely
causes of loss in the reservoir. The tags and tagging method used
in this study have proven reliable in a number of studies already
cited herein, but it cannot be ruled out that the adverse effects of
capture, handling and tagging or transmitter malfunction could
be reasons for the loss of some eels. However, earlier experience
with the tags and studies of tagging effects (Baras and Jeandrain,
1998; Winter et al., 2005) gives reason to believe that it is not a
problem.
A considerable number of eels (n = 22) did not pass the
power station, suggesting that migrating silver eels have
problems in locating the bypass openings. All eels spend a
long time in the area in front of the entrance (power canal), as
seen in the recordings from the station at Site 3 (Table 1). A
number of eels (14) moved in and out of the power canal
several times over a course of days - weeks before finally
passing or disappearing. The opening of the bypass is at the
surface, whereas silver eels migrate in midwater or near the
bottom (Tesch, 2003). Consequently, the eels have to approach
the surface to find the bypass. Haro et al. (2000) observed eels
occupying a deep portion of a forebay, down to 10 m, but the
eels made frequent excursions to the surface. Despite the fact
that eels are known to be bottom-oriented, their vertical
searching behaviour makes it possible for eels to locate and
bypass hydroelectric stations through a surface bypass
and spillway (Durif et al., 2003; Gosset et al., 2005; Watene
and Boube
´e, 2005). The 10 mm bar spacing at the Tange
power station prevents silver eels (down to approximately
35 cm length) from being sucked into the turbines. However,
they can often be found impinged on the turbine screens; the
proportion of eels that suffer this fate can be high (Calles et al.,
2010). Gosset et al. (2005) found impingement to be related to
the flow rate, and suggested a water velocity of less than
0.3 m s
)1
to prevent impingement on 20 mm bar spacing on
racks. The risk of impingement probably depends on the water
Table 1
Number of acoustic tagged silver eels (Anguilla anguilla) detected at downstream sections (Fig. 1), time spent and progression rate of downstream migration in each River Gudena
˚section. Mean length of time
(minutes) spent within range of each detection station is given
River section Release – Site 1 Site 1–2 Site 2–3 Site 3–4 Site 4–5
Habitat River River Reservoir Power station River
No. of eels detected 43 (100%) 43 (100%) 38 (88%) 16 (37%) 10 (23%)
Mean time (h) (range; SD) 226 (4–918; 263) 21 (2–335; 65) 220 (5–881;224) 512 (25–1680; 545) 32 (10–152;43)
Mean progression rate (km h
)1
) (range; SD) 0.24 (0.95–0.00; 0.3) 3.71 (5.25–0.04; 1.4) 0.16 (1.68–0.01; 0.3) 0.04 (0.14–0.00; 0.1) 1.05 (3.26–0.22; 1.1)
Distance from release site (km) 4.1 18.2 26.7 30.2 63.5
Mean time (min) for passing each detection station (range) 4.7 (1–22) 22.9 (1–260) 1179.5 (108–5760) 39.6 (1–600) 2.5 (1–6)
192 M. I. Pedersen et al.
temperature, eels being less likely to escape at low tempera-
tures. Measurement of water velocity in front of the racks at
Tange during low to moderate flow, showed values ranging
from 0.26 to 0.48 m s
)1
. During high flow, when eels are
typically moving, values of up to 1 m s
)1
can be expected. Eels
that impinge on the turbine screens at Tange are subsequently
removed (often dead or severely damaged) from the water with
an automatic debris cleaner. Workers at the power station may
spot eels among debris and subsequently release them down-
stream, or the eels will be trashed with the debris and die. Such
releases of injured eels from the power plant may explain the
relatively large proportion of eels (n = 6) lost in the last river
section.
Combining the results from river loss in the present study
with the loss in the estuary (Aarestrup et al., 2010) demon-
strates a large loss of migrating silver eels in the Gudenaa
system. The loss may be cumulative, with 10% or fewer
A. anguilla reaching the sea. EU management goals for eels are
thus not met in this system and action is required.
References
Aarestrup, K.; Jepsen, N.; Økland, F.; Rasmussen, G., 1999:
Movements of two strains of radio-tagged Atlantic salmon,
Salmo salar L., smolts through a reservoir. Fish. Mgmt. Ecol. 6,
97–107.
Aarestrup, K.; Thorstad, E. B.; Koed, A.; Jepsen, N.; Svendsen, J. C.;
Pedersen, M. I.; Skov, C.; Økland, F., 2008: Survival and
behavior of European silver eel during spring migration in late
freshwater and early marine phase. Fish. Mgmt. Ecol. 15, 435–
440.
Aarestrup, K.; Thorstad, E. B.; Koed, A.; Jepsen, N.; Svendsen, J. C.;
Pedersen, M. I.; Skov, C.; Økland, F., 2010: Survival and
progression rates of large European silver eel Anguilla anguilla
in late freshwater and early marine phase. Aquat. Biol. 9, 263–
270.
Baras, E.; Jeandrain, D., 1998: Evaluation of surgery procedures for
tagging eel Anguilla anguilla (L.) with biotelemetry transmitters.
Hydrobiologica 371 ⁄372,107–111.
Behrmann-Godel, J.; Eckmann, R., 2003: A preliminary telemetry
study of the migration of silver European eel (Anguilla anguilla L.)
in the River Mosel, Germany. Ecol. Freshw. Fish. 12, 196–202.
Boube
´e, J. A. T.; Williams, E. K., 2006: Downstream passage of silver
eels at a small hydroelectric facility. Fish. Mgmt. Ecol. 13(3),165–
176.
Breteler, J. K.; Vriese, T.; Borcherding, J.; Breukelaar, A.; Jo
¨rgensen,
L.; Staas, S.; Laak, G.; Ingedahl, D., 2007: Assessment of
population size and migration routes of silver eel in the River
Rhine based on a 2-year combined mark-recapture and telemetry
study. ICES J. Mar. Sci. 64, 1450–1456.
Calles, O.; Olsson, I. C.; Comoglio, C.; Kemp, P. S.; Blunden, L.;
Schmitz, M.; Greenberg, L. A., 2010: The implication of size-
dependent mortality and decreasing densities of silver eels in
regulated rivers. Freshw. Biol. 55, 2167–2180.
Carr, J. W.; Whoriskey, F. G., 2008: Migration of silver American eels
past a hydroelectric dam and through a coastal zone. Fish. Mgmt.
Ecol. 15, 393–400.
Durif, C.; Elie, P.; Gosset, C.; Rives, J.; Travade, F., 2003: Behavioral
study of downstream migrating eels by radio telemetry at a small
hydroelectric power plant. Am. Fish. Soc. Symp. 33, 343–356.
EU, 2007: Council Regulation (EC) No 1100 ⁄2007 of 18 September
2007 establishing measures for the recovery of the stock of
European eel. Off. J. E. U. L 248, 17–22.
Gosset, C.; Travade, F.; Durif, C.; Rives, J.; Elie, P., 2005: Test of two
types of bypass for downstream migration of eels at a small
hydroelectric power plant. River Res. Appl. 21, 1095–1105.
Haraldstad, Ø.; Vøllestad, L. A.; Jonsson, B., 1984: Descent of
European silver eels, Anguilla anguilla L. in a Norwegian
watercourse. J. Fish Biol. 26, 37–41.
Haro, A.; Castro-Santos, T.; Boube
´e, J., 2000: Behavior and passage of
silver phase American eels at a small hydroelectric facility. Dana
12, 33–42.
ICES, 2001: Report of the EIFAC ⁄ICES Working Group on Eels.
ICES, CM 2001 ⁄ACFM:03, Copenhagen , 87 p.
ICES, 2009: Report of the 2009 session of the Joint EIFAC ⁄ICES
Working Group on Eels. EIFAC Occasional Paper. No. 45. ICES
CM 2009 ⁄ACOM:15.ICES, Rome, FAO ⁄Copenhagen 2010.540
p. (Online).
Maxwell, S. E.; Delaney, H. D., 1990: Designing Experiments and
Analyzing Data: A Model Comparison Perspective. International
Student Ed, Wadsworth, Belmont, Canada.
Monten, E., 1985: Fish and turbines. Fish injury during passage
through power station turbines. Vattenfall, Stockholm.
Potvin, C.; Lechowich, M. J.; Bell, G.; Schoen, D., 1990: The statistical
analysis of ecophysiological response curves obtained from
experiments involving repeated measures. Ecology 71, 1389–1400.
Simon, J.; Fladung, E., 2009: Untersuchungen zur Blankaalabwande-
rung aus Oberhavel, Rhein und Mittelelbe. Fisch. Teichwirt. 60,
288–289.
SPSS, 1997: SPSS Advanced Statistics 7.5, SPSS, Chicago, IL, USA.
Tesch, F. W., 2003: The Eel, Blackwell Science, Oxford, UK.
Vøllestad, L. A.; Jonsson, B.; Hvidsten, N. A.; Næsje, T. F.;
Haraldstad, Ø.; Ruud-Hansen, J., 1986: Environmental factors
regulation the seaward migration of European silver eels (Anguilla
anguilla). Can. J. Fish. Aquat. Sci. 43, 1909–1916.
Von Ende, C. N., 1993: Repeated measures analysis: growth and other
time-dependent measures. In: Design and Analysis of Ecological
Experiments. S. M. Scheiner, J. Gurevitch (Eds), Chapman &
Hall, London, pp. 113–137.
Watene, E. M.; Boube
´e, J. A. T., 2005: Selective opening of
hydroelectric dam spillway gates for downstream migrant eels in
New Zealand. Fish. Mgmt. Ecol. 12, 69–75.
Winter, H. V.; Jansen, H. M.; Adam, B.; Schwevers, U., 2005:
Behaviour effects of surgically implanting transponders in Euro-
pean eel. Anguilla anguilla. In: Aquatic telemetry: advances and
applications. M. T. Spedicato, G. Marmulla, G. Lembo (Eds),
COISPA technoloia & ricerca, Rome, Italy, pp. 1–9.
Winter, H. V.; Jansen, H. M.; Bruijs, M. C. M., 2006: Assessing the
impact of hydropower and fisheries on downstream migration
silver eel, Anguilla anguilla, by telemetry in the River Meuse. Ecol.
Freshw. Fish. 15, 221–228.
Winter, H. V.; Jansen, H. M.; Breukelaar, A. W., 2007: Silver eel
mortality during downstream migration in the River Meuse, from
a population perspective. ICES J. Mar. Sci. 64, 1444–1449.
AuthorÕs address: Michael I. Pedersen, DTU-Aqua, Section for
Freshwater Fisheries Ecology, Vejlsøvej 39, DK-
8600 Silkeborg, Denmark.
E-mail: mip@aqua.dtu.dk
Loss of European silver eels 193