Content uploaded by León Jesús German-Ponciano
Author content
All content in this area was uploaded by León Jesús German-Ponciano on Jun 02, 2022
Content may be subject to copyright.
Content uploaded by Juan F. Rodríguez-Landa
Author content
All content in this area was uploaded by Juan F. Rodríguez-Landa on Jun 01, 2022
Content may be subject to copyright.
Molecules 2022, 27, x. https://doi.org/10.3390/xxxxx www.mdpi.com/journal/molecules
Review
Pharmacological, Neurochemical, and Behavioral Mechanisms
Underlying the Anxiolytic- and Antidepressant-Like Effects of
Flavonoid Chrysin
Juan Francisco Rodríguez-Landa 1,2, *, León Jesús German-Ponciano 1, Abraham Puga-Olguín 3
and Oscar Jerónimo Olmos-Vázquez 1
1 Laboratorio de Neurofarmacología, Instituto de Neuroetología, Universidad Veracruzana,
Xalapa 91190, Mexico; lgerman@uv.mx (L.J.G.-P.); oscarplsstahp1@gmail.com (O.J.O.-V.)
2 Facultad de Química Farmacéutica Biológica, Universidad Veracruzana, Xalapa 91000, Mexico
3 Unidad de Salud Integrativa, Centro de EcoAlfabetización y Diálogo de Saberes, Universidad Veracruzana,
Xalapa 91090, Mexico; abpuga@uv.mx
* Correspondence: juarodriguez@uv.mx
Abstract: Chrysin (5,7-dihydroxyflavone) is a flavonoid isolated from plants, such as Passiflora co-
erulea, Passiflora incarnata, and Matricaria chamomilla. This natural molecule exerts diverse pharma-
cological effects, which includes anti-oxidant, anti-inflammatory, anti-cancer, neuroprotective, and
anti-apoptotic effects. Additionally, in brain structures such as hippocampus, prefrontal cortex, ra-
phe nucleus, and striatum, involved in the physiopathology of anxiety and depression disorders,
several neuropharmacological activities, including the activation of neurotransmitter systems (GA-
BAergic, serotonergic, dopaminergic, and noradrenergic), neurotrophic factors such as brain-de-
rived neurotrophic factor and the nerve growth factor, and some signaling pathways are affected.
The results showed that the anxiolytic and antidepressant-like effects of chrysin occurs through its
interaction with specific neurotransmitter systems, principally the GABAergic and the serotonergic,
and activation of other neurotrophic factors. However, it is not possible to discard the anti-oxidant
and anti-inflammatory activities of chrysin while producing its anxiolytic- and antidepressant-like
effects. Although these results have been obtained principally from pre-clinical research, they con-
sistently demonstrate the potential therapeutic use of flavonoid chrysin as an anxiolytic and anti-
depressant agent. Therefore, this flavonoid could be considered as a promising novel therapy for
anxiety and depression disorders.
Keywords: antidepressant; anxiolytic; chrysin; flavonoid; natural medicine; neuropharmacology
1. Introduction
Throughout the development of pharmacology, the study of natural molecules has
been of special importance to identify secondary metabolites from plants and to stimulate
the discovery and design of new drugs for the treatment of diseases in humans [1]. The
multidisciplinary study of diverse molecules extracted from plants, isolated, and chemi-
cally characterized, has permitted the identification of the mechanism of action involved
in its beneficial and toxic effects [2]. The use of molecular, biochemical, pharmacological,
histological, and behavioral techniques has helped develop multi-target drugs for the pre-
vention and treatment of diverse diseases, particularly those that have recently increased
in association with social dynamics, lifestyle, and environmental factors, such as neuro-
psychiatric and stress-related disorders [3].
Polyphenols are a group of molecules with multiple beneficial effects on health. Its
anti-oxidant effects beneficially impact the physiological process of the organism [4].
Among polyphenols, flavonoids have been widely studied to develop complementary
Citation: Rodríguez-Landa, J.F.; Ger-
mán-Ponciano, L.J.;
Puga-Olguín, A.; Olmos-Vázquez,
O.J. Pharmacological, Neurochemi-
cal, and Behavioral Mechanisms Un-
derlying the
Anxiolytic- and Antidepressant-Like
Effects of Flavonoid Chrysin.
Molecules 2022, 27, x.
https://doi.org/10.3390/xxxxx
Academic Editor(s):
Received: date
Accepted: date
Published: date
Publisher’s Note: MDPI stays neu-
tral with regard to jurisdictional
claims in published maps and institu-
tional affiliations.
Copyright: © 2022 by the authors.
Submitted for possible open access
publication under the terms and con-
ditions of the Creative Commons At-
tribution (CC BY) license (https://cre-
ativecommons.org/licenses/by/4.0/).
Molecules 2022, 27, x FOR PEER REVIEW 2 of 18
therapeutic strategies for the treatment of metabolic, cardiovascular, neuropsychiatric dis-
orders and cancer [5–7]. In vitro and in vivo studies on the pharmacological action of the
flavonoid chrysin have identified multiple effects of it on different systems in the organ-
ism [8–10], including the central nervous system (CNS). Chrysin exerts a neuropharma-
cological effect in brain structures such as the amygdala, hippocampus (HP), prefrontal
cortex (PFC), and raphe nucleus, which are involved in the physiopathology of several
neuropsychiatric disorders, such as anxiety and depression [11–14]. The effects include
activation of the GABAergic system by modulating the GABAA/benzodiazepine receptor
complex [14–17]; changes in serotonin levels and modification in the expression of their
receptors such as 5-HT1A and 5-HT2A in the raphe nucleus and HP [13,18]; and restoration
of dopamine and noradrenaline levels in the CNS [12,19]. In addition, chrysin activates
neurotrophic factors that increase brain-derived neurotrophic factor (BDNF) and nerve
growth factor (NGF) levels, thereby activating signaling pathways in the brain [11,20].
However, anti-inflammatory, anti-apoptotic, and neuroprotective effects should be con-
sidered as potential mechanisms of action involved in their anxiolytic- and antidepres-
sant-like effects, considering that neuroinflammation and apoptotic processes are in-
volved in the pathophysiology of anxiety and depression disorders [21].
In this review, we describe, analyze, and discuss the scientific results from pre-clini-
cal research that report the pharmacological, neurochemical, and behavioral mechanisms
that could underlie the anxiolytic and antidepressant effects of flavonoid chrysin, which
is considered as a promising therapy for anxiety and depression disorders. In addition,
we propose the incorporation of chrysin as a potential anxiolytic and antidepressant drug
in future scientific research, not only for its classical actions on neurotransmission sys-
tems, but also for its anti-oxidant and anti-inflammatory effects, and the activation of neu-
rotrophic factors and the associated signaling pathways. This may contribute to the de-
velopment of specific therapies for anxiety and depression disorders according to their
etiology.
2. Generalities of the Flavonoid Chrysin
Flavonoids are polyphenolic compounds that are present in plants. They produce
pharmacological actions in the peripheral and CNS [9]. They can cross the blood-brain
barrier and interact with several neurotransmission systems and, thereby activating sig-
naling pathways in specific brain structures involved in the physiopathology of anxiety
and depression disorders [10]. In particular, the flavonoid chrysin (5,7-dihydroxyflavone)
has been studied for its anti-oxidant properties; however, its neuropharmacological effects
in specific brain structures involved in the physiopathology of several neuropsychiatric
disorders such as anxiety and depression need to be studied [9,10,14].
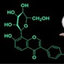
Chrysin (Figure 1) has a backbone structure that consists of a fused A and C rings,
and a phenyl B ring, which is attached to the second position of ring C and shares the
basic structure of the flavones, with an additional hydroxyl group at the fifth and seventh
positions of the A ring. The potential of chrysin to act as a free radical scavenger has been
attributed to the presence of these hydroxyl groups [20,22], and it has been suggested that
these functional groups represent the main site of action of this flavonoid to produce a
great variety of pharmacological activities and therapeutic effects. It has the potential to
be used as an alternative in the treatment of metabolic, cardiovascular, and neuropsychi-
atric disorders [10]. In addition, the presence of hydroxyl groups in the backbone of chry-
sin has been associated with its anxiolytic-like effects [23]. Chrysin, but not the flavone
backbone, decreases anxiety-like behavior in rats and zebrafish, suggesting that the pres-
ence of hydroxyl groups in its basic structure is indispensable for producing anxiolytic-
like effects in pre-clinical research [23].
Molecules 2022, 27, x FOR PEER REVIEW 3 of 18
Figure 1. Basic structure of flavones showing fused A and C rings, and phenyl B rings with corre-
sponding numbering system (left figure). Structure of the flavonoid chrysin, 5,7-dihydroxyflavone
(right figure).
Chrysin, either isolated from plants like Passiflora coerulea, Passiflora incarnata, and
Matricaria chamomilla, or even as a synthetic drug, produces anxiolytic- and antidepres-
sant-like effects. These effects involve several activation of neurotransmission systems
and signaling pathways, including the serotonergic and GABAergic systems, and the ac-
tivation of neurotrophic factors, such as BDNF and NGF. It is likely that the activation of
anti-inflammatory and anti-oxidant signaling pathways may also be involved in these ef-
fects. Although these effects have been principally evaluated in pre-clinical research
[14,16–18,24–26], they show the potential therapeutic use of chrysin for anxiety, depres-
sion, and other neuropsychiatric disorders.
3. Biochemical and Pharmacological Activity of Flavonoid Chrysin
Diverse flavonoids, including chrysin, exert significant anxiolytic- and antidepres-
sant-like activities in mammals and non-mammals, similar to those produced by clinically
effective anxiolytic and antidepressant drugs [5,10,13]. These effects have been related to
its neurochemical activity on the CNS that impact neuronal functioning (Table 1).
Table 1. Some neurochemical effects produced by flavonoid chrysin potentially involved in its an-
xiolytic- and antidepressant-like effects.
Activity
Chrysin Treatment
Effects
Reference
Anti-oxidant
20 mg/kg/30 days, p.o.
↓ TBARS, lipid hydroperoxides, conjugated
dienes tissue, circulatory levels
↑ SOD, CAT, GPx, Gsr activity, GSH, GSTs, vit-
amin C and vitamin E levels in ethanol-induced
toxicity in rats
[22]
50 mg/kg/12 days, i.p.
↑ GHS levels and CAT and SOD activity in
heart homogenate in male rats
[27]
30 and 60 mg/kg/28 days,
p.o.
↑ NO and GHS levels, GSHPx, CAT, and SOD
activity in rat heart homogenate
[28]
60 mg/kg/28 days, p.o
↑ 8-OHdG, TBARS levels
↓ GSH, CAT, NO levels
[29]
1 and 10 mg/kg/60 days,
p.o.
↑ SOD, CAT and GPx activity in PFC and HP of
aged mice
[20]
1.25, 2.5, and 5 µM/30 min
exposure
↓ ROS formation in neuronal SH-SY5Y and mi-
croglial THP-1 cells in vitro
[30]
10, 30, and 100 mg/kg/44
days, p.o.
Protects against aluminum-induced oxidative
stress by restored LPO levels and SOD and
CAT activity in cortex and HP of male Swiss
mice
[30]
Molecules 2022, 27, x FOR PEER REVIEW 4 of 18
Anti-inflammatory
7.50, 4.75, and 120.90 µM,
18 h exposure
↓ NO, PGE2 and TNF-α biosynthesis
in CLP-induced RAW 264.7 cells
[31]
30 mg/kg/2 weeks, i.p.
↓ ALT and AST activity
↓ TNF-α and IL-1β levels
↑ IL-10 and adiponectin in high-fat feeding
mice
[32]
25 and 50 mg/kg/12 days,
i.p.
↓ NF-κB, iNOS, COX-2, and TNF-α expression
in heart homogenate of DOX-induced cardio-
toxicity mice
[27]
30 and 60 mg/kg/28 days,
p.o.
↑ PPAR-γ and TGF-β expression
↓ NF-κBp65 and IKK-β expression
and TNF-α level in heart homogenate of isopro-
terenol-induced myocardial injury rats
[28,29]
5 and 20 mg/kg/28 days,
p.o.
↓ TNF-α, IL-1β and IL-6 levels in PFC and HP
of chronically stressed mice
[18]
5 mg/kg/1 h before LP, i.p.
↓ AST and TNF-α serum levels in septic mice
survival
[33]
5 µM/24 h exposure
↓ iNOS, IL-1β, and TNF-α expression in micro-
glial THP-1 cells exposed to LPS
[30]
GABAergic/BZD
3 µM, 60 min exposure
Acts as competitive ligand for central BZD site
in bovine cerebral cortical membranes in vitro
[15]
13 µM, 60 min exposure
Acts as competitive ligand for peripheral BZD
binding site in rat kidneys
membranes in vitro
[15]
1 mg/kg, i.p.
Activates the GABAA/BZD receptor complex in
male CF1 mice
[24]
1 mg/kg, i.p.
Activates the GABAA/BZD receptor complex in
male Sprague-Dawley rats
[17]
0.62 µM, 2 h exposure
Acts as competitive ligand for central BZD site
in synaptosomal fractions of rat brain in vitro
[34]
10 and 30 µM, 30 s expo-
sure
Modulates the activity of Cl− ion channel in the
GABAA receptor expressed in Xenopus oocytes
in vitro
[35]
2 mg/kg, i.p.
↓ Anxiety-like behavior by modulating Cl− ion
channel in the GABAA receptor of cycling fe-
male rats
[36]
2 mg/kg, i.p.
↓ Depression-like behavior by modulating
GABA-binding site in the GABAA receptor of
ovariectomized female rats
[37]
0.5 µg/rat, i.h.
↓ Anxiety-like behavior by modulating
GABAA/BZD receptor complex in the dorsal
hippocampus of cycling female rats
[14]
Serotonergic
5 and 20 mg/kg, p.o.
↑ 5-HT levels and 5-HIAA/5-HT ratio in HP of
chronic stressed mice
[18]
20 mg/kg/28 days, p.o.
↑ 5-HT levels in PFC and HP in female mice
with hypothyroidism
[12]
50 mg/kg twice a day per 4
days, p.o.
↑ 5-HT levels in the striatum
of the rat brain
[38]
10 and 30 mg/kg/2 weeks,
p.o.
↑ 5-HT spinal levels
[39]
Molecules 2022, 27, x FOR PEER REVIEW 5 of 18
↓ 5-HIAA/5-HT ratio in male mice with experi-
mental neuropathy
5 mg/kg/28 days, i.p.
↓ 5-HT1A receptor expression in
the dorsal raphe
↑ 5-HT1A and 5-HT2A in the hippocampus of
male rats
[13]
Dopaminergic
10 mg/kg/28 days, p.o
↑ DA striatal levels in mice
[19]
50, 100 and 200 mg/kg/ 5
days, p.o.
↑ DA levels in striatum of mice treated
with 1-methyl-1,2,3,6-tetrahidropidine
[40]
20 mg/kg/28 days, p.o.
↑ DA levels in PFC and HP in a hypothyroid-
ism model in female mice
[12]
Noradrenergic
50, 100 and 150 mg/kg, i.p.
↓ NE serum levels in rats with pain
induced by formalin
[41]
20 mg/kg/28 days, p.o.
No effects
[12]
Anti-apoptotic
25 and 50 mg/kg/12 days,
i.p.
↓ Bax, caspase-3, and cytochrome c activity
↑ Bcl-2 expression in rat heart tissue extract
[27]
30 and 60 mg/kg/28 days,
p.o.
↑ Bcl-2 expression
↓ Bax and caspase-3 activity
[29]
5 and 20 mg/kg/28 days,
p.o.
↓ Caspase-3 and caspase-9 activity in HP and
PFC of chronically stressed mice
[18]
25, 50 and 100 mg/kg/3
days, p.o.
↓ Apoptotic index in cerebral cortex
and HP of rats with traumatic brain injury
[42]
Neuroendocrine
5 and 20 mg/kg/28 days,
p.o.
↓ Corticosterone plasma levels in chronically
stressed mice
[11]
5 and 20 mg/kg/28 days,
p.o.
↓ CRH and ACTH in chronically stressed mice
[18]
50, 100 and 150 mg/kg, i.p
↓ Corticosterone serum levels in rats with pain
induced by formalin
[41]
Neurotrophic
5 and 20 mg/kg/28 days,
p.o.
↑ BDNF and NGF levels in PFC and HP in
chronically stressed mice
[11]
1 and 10 mg/kg/60 days,
p.o.
↑ BDNF levels in HP and PFC in aged mice
[20]
10 mg/kg/28 days, p.o.
↑ BDNF and NGF levels in striatum in a Parkin-
son’s disease model in mice
[19]
20 mg/kg/28 days, p.o.
↑ BDNF and NGF in HP and PFC in mice sub-
jected to a hypothyroidism model
[43]
TBARS = thiobarbituric acid reactive substance; SOD = superoxide dismutase; CAT = catalase; GPx
= glutathione peroxidase; Gsr = glutathione reductase; GSTs = glutathione-S-transferase; GSH = re-
duced glutathione; NO = nitric oxide; GSHPx = plasma glutathione peroxidase; 8-OHdG = 8-hy-
droxy-2′-deoxyguanosine; PFC = prefrontal cortex; HP = hippocampus; ROS = reactive oxygen spe-
cies; LPO = lipid peroxidation; PGE2 = prostaglandin E 2; TNF-α = tumor necrosis factor-α; CLP =
cecal ligation and puncture procedure; ALT = alanine aminotransferase; AST = aspartate ami-
notransferase; IL-1β = interleukin 1 beta; IL-10 = interleukin 10; NF-κBp65 = nuclear transcription
116 factor kappa B heterodimer; iNOS = inducible nitric oxide synthase; COX- 2 = cyclooxygenase-
2; DOX = doxorubicin; PPAR-γ = peroxisome proliferator-activated receptor-gamma; TGF-β = trans-
forming growth factor-beta; IKK-β = inhibitor of nuclear factor kappa-B kinase subunit beta; IL-6 =
interleukin-6; LPS = lipopolysaccharide; BZD = benzodiazepine; GABA = gamma-aminobutyric
acid; 5-HT = 5-hydroxytriptamina, serotonin; 5-HIAA = 5-hydroxyindoleacetic; 5-HT1A = serotonin
1A receptor; 5-HT2A = serotonin 2A receptor; DA = dopamine; NE = noradrenaline; BAX = pro-apop-
totic protein of the subfamily Bax; Bcl-2 = B-cell lymphoma-2; CRH = corticotropin-releasing hor-
mone; ACTH = adrenocorticotropic hormone; BDNF = brain-derived neurotrophic factor; NGF =
nerve growth factor; ↑ = increase; ↓ = decrease; p.o. = per oral route; i.p. = intraperitoneal injection;
i.h. = intrahippocampal microinjection.
Molecules 2022, 27, x FOR PEER REVIEW 6 of 18
3.1. Action of Chrysin on Neurotransmission Systems
Chrysin was the first monoflavonoid reported as a specific ligand for central and pe-
ripheral benzodiazepine-binding sites [15]. It was identified when chrysin inhibited the
[3H] flunitrazepam binding in synaptosomal membranes of the bovine cerebral cortex.
Similarly, chrysin also displaced the binding of [3H]Ro 5-4864, a potent ligand for the pe-
ripheral benzodiazepine receptor, to kidney membranes in a mixed, competitive and non-
competitive manner. Interestingly, intracerebroventricular microinjection of chrysin pre-
vents tonic-clonic seizures induced by pentylenetetrazol [15]. This effect was blocked by
Ro 15-1788, a central benzodiazepine receptor antagonist, suggesting that this flavonoid
exerts some of its pharmacological actions on the benzodiazepine-binding site of the
GABAA receptor, enhancing the activation of GABAergic neurotransmission [15]. In sup-
port of this, in vitro studies have reported that chrysin and other flavonoids modulate the
chloride ion channel in the GABAA receptor in a manner different from benzodiazepines
[35].
Additionally, chrysin can also activate other neurotransmitter systems involved in
the physiopathology of several neuropsychiatric disorders, including anxiety and depres-
sion. Chrysin at 10 mg/kg for 28 days restored dopamine, homovanillic acid, and 3,4-di-
hydroxyphenylacetic acid levels in the stratum of male C57B/6J mice, in which a depletion
of this neurotransmitter was induced by 6-hidroxidopamine [19]. In addition, chrysin at
50, 100 and 200 mg/kg for 5 days prevented the decrease in dopamine in the striatum
produced by the injection of 1-methyl-a,2,3,6-tetrahidropidine in C57BL/6J mice [40]. In
contrast, chrysin at 20 mg/kg for 28 days reversed the decrease in dopamine and serotonin
levels in the PFC and HP of female mice subjected to a hypothyroidism model [12]. Chry-
sin at 50 mg/kg for 4 days and chrysin 3–30 mg/kg for 2 weeks significantly increased
serotonin levels in mice and rat brains [38,39]; while chrysin 50, 100, and 150 mg/kg atten-
uated the increased noradrenaline and corticosterone levels in male Wistar rats with pain
induced by formalin [41].
It has been shown that the flavonoid chrysin has a positive impact on several neuro-
transmitter systems that participate in the etiology of anxiety and depression; however, it
is also capable of modifying several signaling pathways and activating neurotrophic pro-
cesses in the CNS, which could be related to its anxiolytic- and antidepressant-like effects.
3.2. Anti-Oxidant Activity of Chrysin
Psychiatric disorders have been linked to alterations in the activity of anti-oxidant
enzymes in the CNS, leading to an increase in the production of reactive oxygen species
(ROS) and, in the first instance, induction of oxidative stress processes [44]. Oxidative
stress leads to an imbalance between the level of anti-oxidants and production of ROS,
which can generate long-term alterations such as damage to neuronal membranes with
potential activation of apoptosis processes associated with the increase of ROS [45]. These
alterations, induced by oxidative stress, are involved in the etiology of anxiety and de-
pression disorders [46,47]. Chrysin has the capacity to modulate the nitric oxide (NO)
pathway, which contributes to the production of ROS, in both in vivo and in vitro assays
[31,33]. In a diabetic rat model, chrysin 60 mg/kg for 28 days inhibited oxidative stress by
restoring the alterations in NO, glutathione (GSH), catalase (CAT), thiobarbituric acid re-
active substances (TBARS) levels, along with the restoration of the activity of superoxide
dismutase (SOD), nitrotyrosine (NT), and NADPH oxidase 4 (Nox4) [29]. The same effect
was produced by the administration of chrysin at 20 mg/kg for 30 days, exerting a reduc-
tion in ethanol-induced toxicity, as evidenced by decreasing markers of oxidative stress
in organs such as the liver, kidney, and heart of rats [22]. In addition, chrysin 50 mg/kg
restored the levels of oxidative stress markers such as GSH, malondialdehyde (MDA),
CAT, SOD, and ROS under doxorubicin-induced acute cardiotoxicity in rats to normal
values [27]. The anti-oxidant effects of chrysin described above occur in the peripheral
organs of metabolically challenged (diabetic) or chemically treated (ethanol and
Molecules 2022, 27, x FOR PEER REVIEW 7 of 18
doxorubicin) animals; which is important because diabetes and alcohol intake is associ-
ated with oxidative stress and inflammation [48,49], and may predispose to anxiety and
depression disorders [50,51]. Interestingly, it has been previously reported in preclinical
and clinical studies that reduced oxidative stress at the peripheral level is associated with
a decrease in anxiety and depression symptoms [52–55]. In this way, it is possible that
peripheral and central anti-oxidant effects of chrysin could contribute to its anxiolytic and
antidepressant effects; however, this aspect needs to be explored further.
In support, daily administration of chrysin at 1 and 10 mg/kg for 60 days restored the
activity of SOD and CAT in the HP and PFC of aged mice and reduced oxidative stress by
decreasing ROS and inversely increasing BDNF levels in these brain structures [20]. This
last data is important because a reduction in anxiety-like behaviors in the elevated plus
maze (EPM) test was related to high levels of BDNF, which was accompanied by greater
expression of TrkB and BDNF mRNA in brain structures such as the HP, PFC, and amyg-
dala [56]. Interestingly, the anxiolytic-like activity produced by diazepam has been asso-
ciated with the restoration of normal levels of anti-oxidant enzymes, such as SOD and
CAT, as well as a reduction in nitrite concentration in the mouse brain [57]. Therefore,
alterations in peripheral and central enzymatic anti-oxidant systems can generate oxida-
tive stress and contribute to anxiety- and depression-like behaviors. In this way, it is pos-
sible that these alterations related to oxidative stress may be restored by the flavonoid
chrysin and contribute to its anxiolytic- and antidepressant-like effects, without discard-
ing the participation of other neurochemical processes in the CNS.
3.3. Anti-Inflammatory and Anti-Apoptotic Activity of Chrysin
In vitro studies with chrysin showed attenuated release of pro-inflammatory media-
tors by inhibiting prostaglandins E2 (PGE2) and tumor necrosis factor-α (TNF-α) from
lipopolysaccharide-induced RAW 264.7 cells [31]. Chrysin regulates inflammatory and
apoptotic signaling pathways by attenuating the effects of nuclear factor-κB (NF-κB) ex-
pression, inhibition of nuclear factor kappa-B kinase (IKK-β), TNF-α, interleukin-1β (IL-
1β), interleukin-6 (IL-6), and cycloxygenase-2 (COX-2) [27,28,32]. In addition, in a mouse
model of polymicrobial sepsis induced by cecal ligation, the administration of chrysin at
5 mg/kg reduced TNF-α levels [33]. In addition, chrysin at 50 and 60 mg/kg for 12 to 28
days in models of myocardial injury in diabetic rats or doxorubicin-induced acute cardi-
otoxicity exerts anti-inflammatory effects. These effects were associated with the inhibi-
tion of TNF-α and NF-κB/IKK-β expression, as well as with the reduced apoptosis due to
increase in Bcl-2 (anti-apoptotic protein) expression and decrease in the expression of pro-
apoptotic markers such as Bax and caspase-3 [27,28]. These effects of chrysin at the pe-
ripheral level and in different models of inflammation support its potential anti-inflam-
matory and anti-apoptotic activities. This is important because peripheral and central pro-
inflammatory processes have been involved in anxiety development and depression dis-
orders [58], while some anxiolytic effects of drugs have been related to stable functioning
of COX-2 in the infralimbic and prelimbic cortex, HP, and ventral tegmental area [59]. In
addition, anxiolytic- and antidepressant-like effects produced while exercising are due to
the increase in Bcl-2 and the reduction of Bax and caspase-3 in the dorsal raphe nucleus of
rats evaluated in the EPM and forced swim test (FST) without observing neuronal death
[60,61]. In contrast, a reduction in Bcl-2 and an increase in Bax in the frontal cortex are
related to anxiety-like behavior and other stress-related disorders [60]. Therefore, chrysin
can produce anxiolytic- and antidepressant-like effects by exerting beneficial effects on
these biochemical systems.
Similarly, chrysin at 5 and 20 mg/kg for 28 days produced significant changes in neu-
rochemical factors that are implicated in the reduction of pro-inflammatory cytokines in
mice subjected to the chronic unpredictable mild stress model (CUMS) [18]. Furthermore,
chrysin also prevented the increase in concentrations of corticotropin-releasing hormone
(CRH) and adrenocorticotropic hormone (ACTH) in plasma, while the levels of TNF-α,
IL-1β, and IL-6 in the PFC and HP were reduced by both doses of chrysin [18]. Similarly,
Molecules 2022, 27, x FOR PEER REVIEW 8 of 18
under these conditions, restoration of caspase-3 and caspase-9 activity in the HP and PFC
was observed. All these changes were associated with a reduction in depression-like be-
haviors, reaching values similar to those reported in unstressed mice [18].
It is necessary to highlight that mice and rats that displayed lower anxiety-like be-
havior in the EPM test showing lower expression of pro-inflammatory cytokines, includ-
ing TNF-α, IL-1β, and IL-6 in the HP, frontal cortex, and serum [44,62]. However, a con-
trary effect was reported in animals showing high anxiety-like behavior. Additionally,
chrysin can regulate the hypothalamic-pituitary-adrenal (HPA) axis and physiological re-
sponses to stressful situations [18]. Adequate regulation of the HPA axis combined with
the lower production and release of corticosterone has been associated with the adequate
coping with stress and low risk of developing neuropsychiatric disorders [63,64].
Altogether, these results suggest that neurochemical changes produced by flavonoid
chrysin, in addition to suppressive effects on inflammatory and oxidative processes, could
contribute to its anxiolytic- and antidepressant-like effects [42,65]. These results support
the idea that chrysin could be considered a potential candidate for ameliorating anxiety
and depression symptoms in humans.
3.4. Effects of Chrysin on Gut-Microbiota
Currently, there is growing evidence proposing that dysregulation in the composi-
tion of the gut-microbiota is related to the pathophysiology of anxiety and depression
disorders due to its interaction with neuroimmune, neuroendocrine, and neural pathways
[66,67]. These pathways are part of the brain–gut–microbiota axis that may modulate
brain development and function, which also impacts on behavior. For example, a fecal
microbiota sample from depressed patients when transferred by oral gavage to a micro-
biota-deficient rat model produced depressive-like behaviors in the recipient animal [68].
Contrarily, fecal microbiota coming from intact rats transplanted to chronically stressed
rats significantly reduced depression- and anxiety-like behaviors and anhedonia, which
was associated with the suppressed activation of glial cells and the NLRP3 inflammasome
in the brain [69]. Interestingly, polyphenols may regulate the composition of the gut-mi-
crobiota, promoting beneficial effects on the intestinal microbiota and inhibiting the pro-
liferation of harmful bacteria, thereby maintaining intestinal health and ameliorating
some neurological and neuropsychic disorders [70,71]; this is important because a correct
structure of gut microbiota in addition to an anti-inflammatory effect of diverse mole-
cules, including polyphenols, has been associated with its antidepressant-like effects [72].
In the specific case of chrysin, its beneficial effects in modulating the structural alteration
of the gut microbiota in mice have been explored [73]. Chrysin 10 mg/kg for 28 days at-
tenuated the increase in pro-inflammatory cytokines and the intestinal damage induced
by LPS in C57BL6/J mice, which was associated with adequate gut microbiota structure
[74]. Likewise, chrysin can modulate intestinal inflammation in Caco2 cells stimulated
with IL-1B, improving intestinal absorption and metabolic stability [75], which could be
related with a normal function of the gut microbiota; this is important because anti-in-
flammatory effects of several molecules at the peripheral and central level have been as-
sociated with a reduction of anxiety and depression-like behavior [58]. In this sense, it is
possible that the regulation of the gut microbiota by polyphenols such as chrysin may
open new perspectives to explore the effects of chrysin on the brain-gut-microbiota axis
and its potential relationship with its anxiolytic- and antidepressant-like effects.
4. Anxiolytic-Like Effects of Flavonoid Chrysin
In 1994, Wolfman et al. reported an anxiolytic-like effect of chrysin in mice. A single
dose of chrysin at 1 mg/kg significantly increased the time spent in open arms of the EPM
[24]. In the light/dark box (LDB), increased time was spent in the illuminated compart-
ment [17], and in both cases the effects were similar to that produced by diazepam. These
behavioral effects are considered to be associated with anxiolytic-like effects in pre-clinical
Molecules 2022, 27, x FOR PEER REVIEW 9 of 18
research. Chrysin, but not diazepam, is devoid of motor effects related to sedation [24];
and this may represent the advantage of chrysin over benzodiazepines such as diazepam
in the treatment of anxiety disorders [76]. Interestingly, in male Sprague-Dawley rats, the
anxiolytic-like effects of chrysin at 1 and 2 mg/kg in LDB were blocked by previous ad-
ministration of flumazenil [17,77], an antagonist of the benzodiazepine binding site in the
GABAA receptor. Additionally, acute administration of chrysin (1 mg/kg) produced anxi-
olytic-like effects in male Wistar rats [78] and CD-1 male mice [79] evaluated in the EPM.
Similarly, anxiolytic-like effects of chrysin in mammals (mice and rats) have also been
reported in non-mammalian organisms (zebrafish). Chrysin at 1 mg/kg decreased anxiety-
like behavior in rats and zebrafish, similar to diazepam [23]; however, treatment with a
flavone backbone at 1 mg/kg was devoid of anxiolytic-like effects in both rats and
zebrafish, suggesting that the presence of hydroxyl groups in its basic structure could be
indispensable to produce anxiolytic-like effects [23].
Anxiety symptoms in women are associated with a reduction in steroid hormones
such as estradiol and progesterone, and its reduced metabolite allopregnanolone in the
peripheral and CNS, which may occur pre-menstruation, post-partum, and transition to
menopause stage [80,81]. These steroid hormones may modulate several neurotransmis-
sion systems, such as the serotoninergic, noradrenergic, dopaminergic, and GABAergic
[82]; therefore, some of these hormones have been proposed as novel groups of anxiolytic
drugs for treating particular anxiety and depression disorders associated with reduced
concentrations of steroid hormones [83]. It has recently been proposed that the flavonoid
chrysin mimics some of the pharmacological effects of neurosteroids in female rats [37].
Anxiety-like behaviors in female rats significantly increase during metestrus-diestrus
phase of the ovarian cycle, which is associated with low concentration of steroid hormones
[84]; this phase is considered an equivalent of the premenstrual period in women [85].
Interestingly, chrysin at 2 mg/kg, similar to diazepam at 2 mg/kg, prevents anxiety-like
behavior that naturally occurs during metestrus-diestrus phase in female rats evaluated
in the EPM and LDB. This effect can be blocked by a previous injection of picrotoxin [36].
In support, microinjection of chrysin at 0.5 μg in the dorsal HP prevented anxiety-like
behavior that naturally occurs during diestrus, which was blocked by previous injection
of picrotoxin, bicuculline, and flumazenil, indicating that the GABA/benzodiazepine re-
ceptor complex in the dorsal HP mediates the anxiolytic-like effects of this flavonoid [86].
Interestingly, this same effect on anxiety-like behavior during diestrus was prevented by
microinjection of neurosteroid allopregnanolone at 0.5 μg into the dorsal HP, which was
blocked by picrotoxin, bicuculline, and flumazenil in the EPM [86]. In contrast, in a surgi-
cal menopause model in rats characterized by high anxiety-like behavior associated with
a permanent reduction of steroid hormones, chrysin at 2 and 4 mg/kg and diazepam at 1
mg/kg, reversed this anxiety-like behavior, which was blocked by a previous injection of
picrotoxin [26]. The fact that picrotoxin, bicuculline, and flumazenil prevented the anxio-
lytic-like effect of different doses of chrysin supports the idea that its pharmacological
effects are established on the GABA/benzodiazepine receptor complex, as occurs with
clinically effective GABAergic anxiolytic drugs and several neurosteroids, such as allo-
pregnanolone [87], but does not produce the typical sedative effects of benzodiazepines
[24]. However, we cannot discard the possibility of other neurotransmitter systems’ par-
ticipation and the anti-inflammatory and anti-oxidant effects in different regions of the
brain due to the anxiolytic-like effects of chrysin (Figure 2). Specific studies are required
to support or discard this possibility.
Molecules 2022, 27, x FOR PEER REVIEW 10 of 18
Figure 2. Mechanism of action of the flavonoid chrysin potentially involved in its anxiolytic-like
effects. (A) It has been confirmed that chrysin produces its anxiolytic-like effect through its action
on the GABAA/benzodiazepine receptor complex producing configurational changes in the receptor
and regulating the opening of the Cl− ion channel [14,15,17,24,35]; which may produce inhibitory
effects in the GABAergic system associated with its anxiolytic-like effects. These effects can be
blocked by specific antagonists of the GABAA receptor such as picrotoxin, bicuculline and fluma-
zenil [14]. (B) Probably, anti-oxidant effects of chrysin could be involved in its anxiolytic-like effects.
Chrysin significantly reduces ROS by inhibiting the production of NO, NT, and NOX4 [29]. These
effects reduce the oxidative stress and reduces the neuronal damage. Additionally, chrysin reduces
the activity of Bax, caspase-9 and caspase-3, while increasing the production of Bcl-2, thereby reduc-
ing the damage of DNA and inhibiting apoptotic processes [60,61]; which reduces the neuronal
death. (C) Additionally, the anti-inflammatory effects of chrysin could contribute in its anxiolytic-
like effects, considering that it may reduce the inflammatory response by inhibiting the signaling
pathway NF-κB/IKK-β [27,28]. Chrysin may attenuate the expression of NF-κB that participates as
transcriptional factors at nuclear level, binding to genes that induce neuro-inflammation process.
Chrysin also inhibits the production of pro-inflammatory cytokines like IL-1β and IL-6, in addition
to suppressing the production of proinflammatory mediators like TNF-α, PGE2 and COX-2
[27,28,32]. These effects could reduce neuro-inflammation associated with the anxiety-like behavior.
ROS = reactive oxygen species; NO = nitric oxide; NT = nitrotyrosine; NOX4 = NADPH oxidase; O2¯
= superoxide; Green circles = chlorine ions; SOD = superoxide dismutase; GSH = reduced glutathi-
one; CAT = catalase; GPx = glutathione peroxidase; Bcl-2 = anti-apoptotic protein of the subfamily
Bcl-2; Bax = pro-apoptotic protein of the subfamily Bax; NF-κB = nuclear factor kappa B; IKK-β =
inhibitor of nuclear factor kappa-B; TNF-α = tumor necrosis factor-α; IL-1β = interleukin-1β; IL-6 =
interleukin-6; PGE2 = prostaglandins E2; COX-2 = cycloxygenase-2. (Figure was prepared by the
authors).
5. Antidepressant-Like Effects of Flavonoid Chrysin
Few studies have explored the antidepressant-like effects of chrysin; however, their
results are promising. Filho et al. [18] reported that chrysin at 5 and 20 mg/kg for 28 days
increased sucrose consumption and decreased immobility in the tail suspension test (TST)
in female mice C57B/6J exposed to CUMS, which is considered to have antidepressant-
like effects in pre-clinical research. This effect was also associated with an increase in ser-
otonin, BDNF, and NGF levels, and decreased pro-inflammatory levels of cytokines such
Molecules 2022, 27, x FOR PEER REVIEW 11 of 18
as TNF-α, IFN-γ, IL-1β, and IL-6 in the HP and PFC of C57B/6J mice [11,18]. Additionally,
chrysin at 20 mg/kg for 14 days produced an antidepressant-like effect in the FST in male
mice C57B/6J subjected to depression induced by olfactory bulbectomy. This effect was
associated with decreased pro-inflammatory cytokines (i.e., TNF-α, IFN-γ, IL-1β, IL-6),
kynurenine (KYN, a metabolite resulting from serotonin degradation), and indolamine-2,
3-dyoxigenase (IDO, enzyme responsible for serotonin metabolism) activity, besides pro-
ducing an increase in BDNF and serotonin levels in HP [25]. Interestingly, chrysin at 1, 5
and 10 mg/kg for 28 days produced antidepressant-like effects in the FST in male Wistar
rats [13]. In addition, chrysin at 1 and 5 mg/kg for 28 days significantly reduced 5-HT1A
receptor expression in the raphe nucleus and increased it in HP, whereas 5-HT2A receptor
expression was increased in HP [13]. These effects were similar to those produced by the
antidepressant fluoxetine at 1 mg/kg for 28 days. In another study, chrysin at 20 mg/kg
for 28 days produced antidepressant-like effects in the TST and FST in female C57BL/6
mice exposed to a depression model induced by hypothyroidism, which were associated
with increased serotonin and dopamine levels in the HP [12].
As previously mentioned, a reduced concentration of ovarian hormones in women,
during their transition to menopause, increases the risk of developing anxiety and depres-
sion symptoms [88]. Interestingly, using a surgical menopause model in Wistar rats, it was
reported that chrysin at 1 mg/kg reversed depression-like behavior in the FST; this effect
was similar to that produced by neurosteroids progesterone at 1 mg/kg and allopregnano-
lone at 1 mg/kg [37]. The effects of chrysin and neurosteroids were blocked by previous
administration of bicuculline, a selective competitive antagonist of the binding site of γ-
aminobutyric acid in the GABAA receptor, which supports the idea that activation of the
GABAergic system participates in the antidepressant-like effect of chrysin, as has been
reported with neurosteroids [89].
Based on the results described above, we suggest that the mechanism of action un-
derlying the antidepressant-like effect of chrysin involves multiple neurochemical pro-
cesses, such as the activation of neurotransmitter systems, anti-inflammatory and anti-
oxidant processes, and the activation of neurotrophic factors (Figure 3); however, further
exploration is required to improve our understanding of these mechanisms underlying
the antidepressant-like effects of chrysin, and to explore its effects in controlled clinical
trials.
Molecules 2022, 27, x FOR PEER REVIEW 12 of 18
Figure 3. Possible mechanisms of action involved in the antidepressant-like effect of chrysin. (A)
The flavonoid chrysin can modulate ERα and ERβ of membrane, which triggers the MAPK/ERK1/2
signaling pathway involved in phosphorylation and subsequently CREB activation (↑CREB), which
promotes the increase of BDNF levels ( ↑BDNF) [11,19,20,43]; which can further activate the
MAPK/ERK1/2 signaling by the TrkB interaction [56]. The above-mentioned pathway also promotes
an increase of TpOH expression (↑TpOH) and serotonin levels (↑Serotonin) resulting in the antide-
pressant-like effect [12,25]. (B) Furthermore, chrysin can decrease the pro-inflammatory cytokine
levels (↓TNF-α, ↓IL-1β, ↓IL-6, ↓IFN-γ), which inhibits IDO activity (↓IDO) improving serotonergic
neurotransmission and producing its antidepressant-like effect [25]. ER = estrogen receptor; MAPK
= mitogen-activated-protein-kinases; CREB = cAMP response element binding; BDNF = brain de-
rived neurotrophic factor; TrkB = tropomyosinreceptor kinase B; TpOH = tryptophan-hydroxylase;
TNF-α = tumor necrosis factor-α; IL-1β = interleukin 1 beta; IL-6 = interleukin 6; IFN-γ = interferon
gamma; IDO = indoleamine 2,3-dioxygenase; ERK1/2 = extracellular signal-regulated kinase 1 and
2; KYN = kynurenine. (Figure was prepared by the authors).
6. Future Considerations
It is noteworthy that human studies focused specifically on the anxiolytic and anti-
depressant properties of the flavonoid chrysin are nonexistent. Despite this, the results
from pre-clinical studies are promising and support the potential therapeutic effects of
chrysin, which are similar to those produced by clinically effective anxiolytic and antide-
pressants drugs. However, unlike other drugs chrysin does not produce side effects on
motor activity associated with sedation. These results from in vitro and in vivo studies in
preclinical research support the feasibility to evaluate the potential anxiolytic and
Molecules 2022, 27, x FOR PEER REVIEW 13 of 18
antidepressant effects of the flavonoid chrysin in human patients. In the future, it could
play a role in developing new pharmacological strategies to ameliorate the symptoms of
anxiety and depression disorders in patients nonrespondent to conventional antidepres-
sant and anxiolytic drugs.
For many years, increased monoamine availability in the synaptic cleft has been hy-
pothesized as the mechanism underlying the therapeutic effects of clinically effective an-
tidepressant drugs [90]. A wide range of antidepressant drugs has been developed based
on this hypothesis [90]. This is a relevant key that supports the potential antidepressant
actions of chrysin, considering that this flavonoid can increase serotoninergic and dopa-
minergic neurotransmission, which is associated with its antidepressant-like effect [18,25].
In addition, activation of the GABAergic system by this flavonoid supports its potential
anxiolytic properties.
Currently, it is known that an increase in BDNF levels is one of the main effects of
antidepressant drugs and significantly contributes to their therapeutic effects [91]. Ac-
cordingly, a relatively recent meta-analysis found higher serum BDNF concentrations in
patients with major depression disorder after 4 to 12 weeks of antidepressant treatment
with a selective serotonin reuptake inhibitor and a selective noradrenaline recapture in-
hibitor [92]. Similarly, treatment with vortioxetine (an inhibitor of the serotonin trans-
porter; 5–15 mg over 4 weeks) increased the plasma BDNF levels in patients with major
depression disorder, based on their basal values [93]. This is important, considering that
chrysin is similar to conventional antidepressants which increases the concentration of
this neurotrophin in pre-clinical research [11].
Neuroinflammation has also been shown to play a crucial role in the risk of neuro-
psychiatric disorders, including anxiety and depression [58,65]. In agreement with this,
some meta-analyses have reported that administration of clinically effective antidepres-
sant drugs, such as selective serotonin reuptake inhibitors, selective noradrenaline
reuptake inhibitors, and tricyclic antidepressants drugs, decrease the pro-inflammatory
cytokine levels (IL-1β, IL-6, and TNFα) in patients diagnosed with major depression dis-
order [94–96]. Pre-clinical research has also demonstrated that chrysin can reduce pro-
inflammatory cytokine levels [11,18,25], which are positively correlated with depression-
like behavior. Similarly, oxidative stress has been shown to be involved in the risk of anx-
iety and depression disorders [46,47], and the anti-oxidant effects of chrysin identified in
pre-clinical research may contribute to its anxiolytic and antidepressant effects.
Finally, as mentioned above, the different neurochemical changes associated with
chrysin treatment may play a significant role in the establishment of anxiolytic- and anti-
depressant-like effects of chrysin, which could be helpful in the treatment of particular
groups of patients. Therefore, in future studies, it will be important to explore the anxio-
lytic and antidepressant effects of chrysin in particular groups of subjects, considering the
etiology of anxiety and depression symptoms. This could help identify specific groups of
patients in which chrysin could be used as an alternative for the treatment of anxiety and
depression disorders, where their etiology could be related to changes in steroid hor-
mones, neurotransmitters, oxidative stress, or neuro-inflammatory processes. It could
permit the evaluation of therapeutic effects of chrysin in human patients, as it has oc-
curred, for example, with the neurosteroid allopregnanlone [83,87,97,98]; and the mecha-
nism of action underlying its anxiolytic and antidepressant effects is shared with those
produced by the flavonoid chrysin.
7. Conclusions
Chrysin exerts diverse pharmacological effects on the peripheral and CNS. Its action
is in particular on some neurotransmitter systems, by activating neurotrophic factors, reg-
ulating biomarkers of oxidative stress, and regulating inflammatory and apoptotic signal-
ing pathways, which contributes to the anxiolytic- and antidepressant-like effects of this
flavonoid. Although these effects have been evaluated principally in mice and rats, the
results are solid and convincing, and could contribute to clinical evaluation of its potential
Molecules 2022, 27, x FOR PEER REVIEW 14 of 18
anxiolytic and antidepressant effects in particular groups of patients in a short time. In
summary, chrysin is a natural molecule that could become a novel and promising com-
plementary therapy for anxiety and depression disorders in humans.
Author Contributions: J.F.R.-L. and L.J.G.-P. conceived the idea of the paper and developed its
structure. All authors selected and discussed the material to be included in the paper. A.P.-O. and
O.J.O.-V. wrote the first draft of the manuscript. All authors have read and agreed to the published
version of the manuscript.
Funding: This study is part of SIREI project no. DGI: 266502021159 to JFR-L. This research was par-
tially funded by Sistema Nacional de Investigadores: Exp. 32753 (J.F.R-L.), 84949 (L.J.G.-P.), 153293
(A.P.-O.).
Institutional Review Board Statement: Not applicable.
Informed Consent Statement: Not applicable.
Data Availability Statement: Not applicable.
Conflicts of Interest: The authors declare that the research was conducted in the absence of any
commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Ferreira, M.J.U. Natural products in drug discovery and human health. Phytochem. Rev. 2021, 20, 1–4.
https://doi.org/10.1007/s11101-020-09736-y.
2. Tresina, P.S.; Selvam, M.S.; Rajesh, A.; Doss, A.; Mohan, V.R. Natural products in drug discovery: Approaches and develop-
ment. J. Pharm. Res. Int. 2021, 33, 93–110. http://doi.org/10.9734/jpri/2021/v33i35A31879.
3. Dzobo, K. The role of natural products as sources of therapeutic agents for innovative drug discovery. Ref. Module Biomed. Sci.
2021, B978-0-12-820472-6.00041-4. https://doi.org/10.1016/B978-0-12-820472-6.00041-4.
4. Bertelli, A.; Biagi, M.; Corsini, M.; Baini, G.; Cappellucci, G.; Miraldi, E. Polyphenols: From theory to practice. Foods 2021, 10,
2595. https://doi.org/10.3390/foods10112595.
5. German-Ponciano, L.J.; Rosas-Sánchez, G.U.; Rivadeneyra-Domínguez, E.; Rodríguez-Landa, J.F. Advances in the preclinical
study of some flavonoids as potential antidepressant agents. Scientifica 2018, 2018, 2963565.
https://doi.org/10.1155/2018/2963565.
6. Fernández-Demeneghi, R.; Rodríguez-Landa, J.F.; Guzmán-Gerónimo, R.I.; Acosta-Mesa, H.G.; Meza-Alvarado, E.; Vargas-Mo-
reno, I.; Herrera-Meza, S. Effect of blackberry juice (Rubus fruticosus L.) on anxiety-like behaviour in Wistar rats. Int. J. Food Sci.
Nutr. 2018, 70, 856–867. https://doi.org/10.1080/09637486.2019.1580680.
7. Safe, S.; Jayaraman, A.; Chapkin, R.S.; Howard, M.; Mohankumar, K.; Shrestha, R. Flavonoids: Structure-function and mecha-
nisms of action and opportunities for drug development. Toxicol. Res. 2021, 37, 147–162. https://doi.org/10.1007/s43188-020-
00080-z.
8. Garg, A.; Chaturvedi, S. A comprehensive review on chrysin: Emphasis on molecular targets, pharmacological actions and bio-
pharmaceutical aspects. Curr. Drug Targets. 2022, 23, 420–436. https://doi.org/10.2174/1389450122666210824141044.
9. Mishra, A.; Mishra, P.S.; Bandopadhyay, R.; Khurana, N.; Angelopoulou, E.; Paudel, Y.N.; Piperi, C. Neuroprotective potential
of chrysin: Mechanistic insights and therapeutic potential for neurological disorders. Molecules 2021, 26, 6456.
https://doi.org/10.3390/molecules26216456.
10. Talebi, M.; Talebi, M.; Farkhondeh, T.; Kopustinskiene, D.M.; Simal-Gandara, J.; Bernatoniene, J.; Samarghandian, S. An up-
dated review on the versatile role of chrysin in neurological diseases: Chemistry, pharmacology, and drug delivery approaches.
Biomed. Pharm. 2021, 141, 111906. https://doi.org/10.1016/j.biopha.2021.111906.
11. Filho, C.B.; Jesse, C.R.; Donato, F.; Giacomeli, R.; Del Fabbro, L.; da Silva Antunes, M.; de Gomes, M.G.; Goes, A.T.R.; Boeira,
S.P.; Prigol, M.; et al. Chronic unpredictable mild stress decreases BDNF and NGF levels and Na+,K+-ATPase activity in the
hippocampus and prefrontal cortex of mice: Antidepressant effect of chrysin. Neuroscience 2015, 289, 367–380.
https://doi.org/10.1016/j.neuroscience.2014.12.048.
12. Bortolotto, V.C.; Pinheiro, F.C.; Araujo, S.M.; Poetini, M.R.; Bertolazi, B.S.; de Paula, M.T.; Meichtry, L.B.; de Almeida, F.P.; de
Freitas Couto, S.; Jesse, C.R.; et al. Chrysin reverses the depressive-like behavior induced by hypothyroidism in female mice by
regulating hippocampal serotonin and dopamine. Eur. J. Pharmacol. 2018, 822, 78–84. https://doi.org/10.1016/j.ejphar.2018.01.017.
13. Germán-Ponciano, L.J.; Rosas-Sánchez, G.U.; Ortiz-Guerra, S.I.; Soria-Fregozo, C.; Rodríguez-Landa, J.F. Effects of chrysin on
mRNA expression of 5-HT1A and 5-HT2A receptors in the raphe nuclei and hippocampus. Rev. Bras. Farmacog. 2021, 31, 352–360.
https://doi.org/10.1007/s43450-021-00164-3.
14. Rodríguez-Landa, J.F.; Hernández-López, F.; Martínez-Mota, L.; Scuteri, D.; Bernal-Morales, B.; Rivadeneyra-Domínguez, E.
GABAA/benzodiazepine receptor complex in the dorsal hippocampus mediates the effects of chrysin on anxiety-like behaviour
in female rats. Front. Behav. Neurosci. 2022, 15, 789557. https://doi.org/10.3389%2Ffnbeh.2021.789557.
Molecules 2022, 27, x FOR PEER REVIEW 15 of 18
15. Medina, J.H.; Paladini, A.C.; Wolfman, C.; Levi de Stein, M.; Calvo, D.; Diaz, L.E.; Peña, C. Chrysin (5,7-di-OH-flavone), a nat-
urally-occurring ligand for benzodiazepine receptors, with anticonvulsant properties. Biochem. Pharmacol. 1990, 40, 2227–2231.
http://doi.org/10.1016/0006-2952(90)90716-x.
16. Salgueiro, J.B.; Ardenghi, P.; Dias, M.; Ferreira, M.B.; Izquierdo, I.; Medina, J.H. Anxiolytic natural and synthetic flavonoid
ligands of the central benzodiazepine receptor have no effect on memory tasks in rats. Pharmacol. Biochem. Behav. 1997, 58, 887-
891. https://doi.org/10.1016/s0091-3057(97)00054-3.
17. Zanoli, P.; Avallone, R.; Baraldi, M. Behavioral characterisation of the flavonoids apigenin and chrysin. Fitoterapia 2000, 71,
S117–S123. https://doi.org/10.1016/S0367-326X(00)00186-6.
18. Filho, C.B.; Jesse, C.R.; Donato, F.; Del Fabbro, L.; de Gomes, M.G.; Goes, A.T.R.; Souza, L.C.; Giacomeli, R.; Antunes, M.; Lu-
chese, C.; et al. Neurochemical factors associated with the antidepressant-like effect of flavonoid chrysin in chronically stressed
mice. Eur. J. Pharmacol. 2016, 791, 284–296. https://doi.org/10.1016/j.ejphar.2016.09.005.
19. Goes, A.T.; Jesse, C.R.; Antunes, M.S.; Ladd, F.V.L.; Ladd, A.A.L.; Luchese, C.; Paroul, N.; Boeira, S.P. Protective role of chrysin
on 6-hydroxydopamine-induced neurodegeneration a mouse model of Parkinson's disease: Involvement of neuroinflammation
and neurotrophins. Chem. Biol. Interact. 2018, 279, 111–120. https://doi.org/10.1016/j.cbi.2017.10.019.
20. Souza, L.C.; Antunes, M.S.; Filho, C.B.; Del Fabbro, L.; de Gomes, M.G.; Goes, A.T.; Donato, F.; Prigol, M.; Boeira, S.P.; Jesse,
C.R. Flavonoid chrysin prevents age-related cognitive decline via attenuation of oxidative stress and modulation of BDNF levels
in aged mouse brain. Pharmacol. Biochem. Behav. 2015, 134, 22–30. https://doi.org/10.1016/j.pbb.2015.04.010.
21. Feiger, J.A.; Snyder, R.L.; Walsh, M.J.; Cissne, M.; Cwiek, A.; Al-Momani, S.I.; Chiou, K.S. The role of neuroinflammation in
neuropsychiatric disorders following traumatic brain injury: A systematic review. J. Head Trauma Rehabil. 2022, in press.
https://doi.org/10.1097/HTR.0000000000000754.
22. Sathiavelu, J.; Senapathy, G.J.; Devaraj, R.; Namasivayam, N. Hepatoprotective effect of chrysin on prooxidant-antioxidant sta-
tus during ethanol-induced toxicity in female albino rats. J. Pharm. Pharmacol. 2009, 61, 809–817.
https://doi.org/10.1211/jpp.61.06.0015.
23. German-Ponciano, L.J.; Costa, B.P.D.; Feitosa, L.M.; dos Santos Campos, K.; da Silva Chaves, S.N.; Cueto-Escobedo, J.; Max-
imino, C. Chrysin, but not flavone backbone, decreases anxiety-like behavior in animal screens. Neurochem. Int. 2020, 140,
104850. https://doi.org/10.1016/j.neuint.2020.104850.
24. Wolfman, C.; Viola, H.; Paladini, A.; Dajas, F.; Medina, J.H. Possible anxiolytic effects of chrysin, a central benzodiazepine
receptor ligand isolated from Passiflora Coerulea. Pharmacol. Biochem. Behav. 1994, 47, 1–4. http://doi.org/10.1016/0091-
3057(94)90103-1.
25. Filho, C.B.; Jesse, C.R.; Donato, F.; Del Fabbro, L.; Gomes de Gomes, M.; Rossito Goes, A.T.; Souza, L.C.; Boeira, S.P. Chrysin
promotes attenuation of depressive-like behavior and hippocampal dysfunction resulting from olfactory bulbectomy in mice.
Chem. Biol. Interact. 2016, 260, 154–162. https://doi.org/10.1016/j.cbi.2016.11.005.
26. Rodríguez-Landa, J.F.; Hernández-López, F.; Cueto-Escobedo, J.; Herrera-Huerta, E.V.; Rivadeneyra-Domínguez, E.; Bernal-
Morales, B.; Romero-Avendaño, E. Chrysin (5,7-dihydroxyflavone) exerts anxiolytic-like effects through GABAA receptors in a
surgical menopause model in rats. Biomed. Pharmacother. 2019, 109, 2387–2395. https://doi.org/10.1016/j.biopha.2018.11.111.
27. Mantawy, E.M.; El-Bakly, W.M.; Esmat, A.; Badr, A.M.; El-Demerdash, E. Chrysin alleviates acute doxorubicin cardiotoxicity
in rats via suppression of oxidative stress, inflammation and apoptosis. Eur. J. Pharmacol. 2014, 728, 107–118.
https://doi.org/10.1016/j.ejphar.2014.01.065.
28. Rani, N.; Bharti, S.; Bhatia, J.; Tomar, A.; Nag, T.C.; Ray, R.; Arya, D.S. Inhibition of TGF-β by a novel PPAR-γ agonist, chrysin,
salvages β-receptor stimulated myocardial injury in rats through MAPKs-dependent mechanism. Nutr. Metab. 2015, 12, 11.
https://doi.org/10.1186/s12986-015-0004-7.
29. Rani, N.; Bharti, S.; Bhatia, J.; Nag, T.C.; Ray, R.; Arya, D.S. Chrysin, a PPAR-γ agonist improves myocardial injury in diabetic
rats through inhibiting AGE-RAGE mediated oxidative stress and inflammation. Chem. Biol. Interact. 2016, 250, 59–67.
https://doi.org/10.1016/j.cbi.2016.03.015.
30. Campos, H.M.; da Costa, M.; da Silva Moreira, L.K.; da Silva Neri, H.F.; Branco da Silva, C.R.; Pruccoli, L.; Dos Santos, F.C.A.;
Costa, E.A.; Tarozzi, A.; Ghedini, P.C. Protective effects of chrysin against the neurotoxicity induced by aluminium: In Vitro and
In Vivo studies. Toxicology 2022, 465, 153033. https://doi.org/10.1016/j.tox.2021.153033.
31. Harasstani, O.A.; Moin, S.; Tham, C.L.; Liew, C.Y.; Ismail, N.; Rajajendram, R.; Harith, H.H.; Zakaria, Z.A.; Mohamad, A.S.;
Sulaiman, M.R.; et al. Flavonoid combinations cause synergistic inhibition of proinflammatory mediator secretion from lipo-
polysaccharide-induced RAW 264.7 cells. Inflamm. Res. 2010, 59, 711–721. https://doi.org/10.1007/s00011-010-0182-8.
32. Feng, X.; Qin, H.; Shi, Q.; Zhang, Y.; Zhou, F.; Wu, H.; Ding, S.; Niu, Z.; Lu, Y.; Shen, P. Chrysin attenuates inflammation by
regulating M1/M2 status via activating PPARγ. Biochem. Pharmacol. 2014, 89, 503–514. https://doi.org/10.1016/j.bcp.2014.03.016.
33. Harasstani, O.A.; Tham, C.L.; Israf, D.A. Kaempferol and chrysin synergies to improve septic mice survival. Molecules 2017, 22,
92. https://doi.org/10.3390/molecules22010092.
34. Wang, H.; Hui, K.M.; Chen, Y.; Xu, S.; Wong, J.T.; Xue, H. Structure-activity relationships of flavonoids, isolated from Scutellaria
baicalensis, binding to benzodiazepine site of GABAA receptor complex. Planta Med. 2002, 68, 1059–1062.
https://doi.org/10.1055/s-2002-36357.
35. Goutman, J.D.; Waxemberg, M.D.; Doñate-Oliver, F.; Pomata, P.E.; Calvo, D.J. Flavonoid modulation of ionic currents mediated
by GABAA and GABAc receptors. Eur. J. Pharmacol. 2003, 461, 79–87. https://doi.org/10.1016/s0014-2999(03)01309-8.
Molecules 2022, 27, x FOR PEER REVIEW 16 of 18
36. Rodríguez-Landa, J.F.; Guillén-Ruiz, G.; Hernández-López, F.; Cueto-Escobedo, J.; Rivadeneyra-Domínguez, E.; Bernal-Mo-
rales, B.; Herrera-Huerta, E.V. Chrysin reduces anxiety-like behavior through actions on GABAA receptors during metestrus-
diestrus in the rat. Behav. Brain Res. 2021, 397, 112952. https://doi.org/10.1016/j.bbr.2020.112952.
37. Cueto-Escobedo, J.; Andrade-Soto, J.; Lima-Maximino, M.; Maximino, C.; Hernández-López, F.; Rodríguez-Landa, J.F. Involve-
ment of GABAergic system in the antidepressant-like effects of chrysin (5, 7-dihydroxyflavone) in ovariectomized rats in the
forced swim test: Comparison with neurosteroids. Behav. Brain Res. 2020, 386, 112590. https://doi.org/10.1016/j.bbr.2020.112590.
38. Haider, M.; Salman, M.; Kaushik, P.; Bharadwaj, N.; Aggarwal, N.B.; Tabassum, H.; Parvez, S. Chrysin ameliorates 3 nitro-
propinoic acid induced neurotoxicity targeting behavioural, biochemical and histological alterations. Int. J. Neurosci. 2020, 1–9.
https://doi.org/10.1080/00207454.2020.1821677.
39. Wu, J.; Wang, Y.; Cui, W.; Zhou, W.; Zhao, X. 5-HT1A receptor-mediated attenuation of heat hyperalgesia and mechanical allo-
dynia by chrysin in mice with experimental mononeuropathy. Reg. Anesth. Pain Med. 2020, 45, 610–619.
https://doi.org/10.1136/rapm-2020-101472.
40. Krishnamoorthy, A.; Sevanan, M.; Mani, S.; Balu, M.; Balaji, S.; Ramajayan, P. Chrysin restores MPTP induced neuroinflamma-
tion, oxidative stress and neurotrophic factors in an acute Parkinson’s disease mouse model. Neurosci. Lett. 2019, 709, 134382.
https://doi.org/10.1016/j.neulet.2019.134382.
41. Farkhondeh, T.; Samarghandian, S.; Azimin-Nezhad, M.; Samini, F. Effect of chrysin on nociception in formalin test and serum
levels of noradrenalin and corticosterone in rats. Int. J. Clin. Exp. Med. 2015, 8, 2465.
42. Rashno, M.; Ghaderi, S.; Nesari, A.; Khorsandi, L.; Farbood, Y.; Sarkaki, A. Chrysin attenuates traumatic brain injury-induced
recognition memory decline, and anxiety/depression-like behaviors in rats: Insights into underlying mechanisms. Psychophar-
macology 2020, 237, 1607–1619. https://doi.org/10.1007/s00213-020-05482-3.
43. Bortolotto, V.C.; Araujo, S.M.; Pinheiro, F.C.; Poetini, M.R.; Meichtry, L.B.; Fronza, M.G.; Boeira, S.P.; Savegnago, L.; Prigol, M.
Chrysin restores memory deficit in hypothyroidism mice: Behavioral, neurochemical and computational approaches involving
the neurotrophinergic system. J. Psychiatr. Res. 2021, 144, 225–233. https://doi.org/10.1016/j.jpsychires.2021.10.018.
44. Gargouri, B.; Bhatia, H.S.; Bouchard, M.; Fiebich, B.L.; Fetoui, H. Inflammatory and oxidative mechanisms potentiate bifenthrin-
induced neurological alterations and anxiety-like behavior in adult rats. Toxicol. Lett. 2018, 294, 73–86.
https://doi.org/10.1016/j.toxlet.2018.05.020.
45. Gandhi, S.; Abramov, A.Y. Mechanism of oxidative stress in neurodegeneration. Oxid. Med. Cell Longev. 2012, 2012, 428010.
https://doi.org/10.1155/2012/428010.
46. Smaga, I.; Niedzielska, E.; Gawlik, M.; Moniczewski, A.; Krzek, J.; Przegaliński, E.; Pera, J.; Filip, M. Oxidative stress as an
etiological factor and a potential treatment target of psychiatric disorders. Part 2. Depression, anxiety, schizophrenia and au-
tism. Pharmacol. Rep. 2015, 67, 569–580. https://doi.org/10.1016/j.pharep.2014.12.015.
47. Bhatt, S.; Nagappa, A.N.; Patil, C.R. Role of oxidative stress in depression. Drug Discov. Today 2020, 25, 1270–1276.
https://doi.org/10.1016/j.drudis.2020.05.001.
48. Di Sarno, R.; Brigida, A.; Caprio, G.G.; Ciardiello, D.; Dallio, M.; Sangineto, M.; Fagoonee, S.; Abenavoli, L.; Luzza, F.; Gravina,
A.G.; et al. Critical review on the use and abuse of alcohol. When the dose makes the difference. Minerva Med. 2020, 111, 344–
353. https://doi.org/10.23736/S0026-4806.20.06584-2.
49. Bhatti, J.S.; Sehrawat, A.; Mishra, J.; Sidhu, I.S.; Navik, U.; Khullar, N.; Kumar, S.; Bhatti, G.K.; Reddy, P.H. Oxidative stress in
the pathophysiology of type 2 diabetes and related complications: Current therapeutics strategies and future perspectives. Free
Radic. Biol. Med. 2022, 184, 114–134. https://doi.org/10.1016/j.freeradbiomed.2022.03.019.
50. Hovatta, I.; Juhila, J.; Donner, J. Oxidative stress in anxiety and comorbid disorders. Neurosci. Res. 2010, 68, 261–275.
Htpps://doi.org/10.1016/j.neures.2010.08.007.
51. Sarwar, H.; Rafiqi, S.I.; Ahmad, S.; Jinna, S.; Khan, S.A.; Karim, T.; Qureshi, O.; Zahid, Z.A.; Elhai, J.D.; Levine, J.C.; et al. Hy-
perinsulinemia associated depression. Clin. Med. Insights Endocrinol. Diabetes. 2022, 15, 11795514221090244.
Htpps://doi.org/10.1177/11795514221090244.
52. Bouayed, J.; Rammal, H.; Younos, C.; Soulimani, R. Positive correlation between peripheral blood granulocyte oxidative status
and level of anxiety in mice. Eur. J. Pharmacol. 2007, 564, 146–149. https://doi.org/10.1016/j.ejphar.2007.02.055.
53. Salim, S.; Sarraj, N.; Taneja, M.; Saha, K.; Tejada-Simon, M.V.; Chugh, G. Moderate treadmill exercise prevents oxidative stress-
induced anxiety-like behavior in rats. Behav. Brain Res. 2010, 208, 545–552. https://doi.org/10.1016/j.bbr.2009.12.039.
54. Kaufmann, F.N.; Gazal, M.; Mondin, T.C.; Cardoso, T.A.; Quevedo, L.Á .; Souza, L.D.; Ghisleni, G. Cognitive psychotherapy
treatment decreases peripheral oxidative stress parameters associated with major depression disorder. Biol. Psychol. 2015, 110,
175–181. https://doi.org/10.1016/j.biopsycho.2015.08.001.
55. Moccia, M.; Capacchione, A.; Lanzillo, R.; Carbone, F.; Micillo, T.; Perna, F.; Brescia Morra, V. Coenzyme Q10 supplementation
reduces peripheral oxidative stress and inflammation in interferon-β1a-treated multiple sclerosis. Ther. Adv. Neurol. Disord.
2019, 12, 1756286418819074. https://doi.org/10.1177%2F1756286418819074.
56. Nakagawa, Y.; To, M.; Saruta, J.; Yamamoto, Y.; Yamamoto, T.; Shimizu, T.; Kamata, Y.; Matsuo, M.; Tsukinoki, K. Effect of
social isolation stress on saliva BDNF in rat. J. Oral Sci. 2019, 61, 516–520. https://doi.org/10.2334/josnusd.18-0409.
57. Khurana, K.; Bansal, N. Lacidipine attenuates caffeine-induced anxiety-like symptoms in mice: Role of calcium-induced oxido-
nitrosative stress. Pharmacol. Rep. 2019, 71, 1264–1272. https://doi.org/10.1016/j.pharep.2019.07.008.
Molecules 2022, 27, x FOR PEER REVIEW 17 of 18
58. Risbrough, V.B.; Vaughn, M.N.; Friend, S.F. Role of inflammation in traumatic brain injury-associated risk for neuropsychiatric
disorders: State of the evidence and where do we go from here. Biol. Psychiatry. 2022, 91, 438–448. https://doi.org/10.1016/j.bi-
opsych.2021.11.012.
59. Zorzo, C.; Méndez-López, M.; Méndez, M.; Arias, J.L. Adult social isolation leads to anxiety and spatial memory impairment:
Brain activity pattern of COx and c-Fos. Behav. Brain Res. 2019, 365, 170–177. https://doi.org/10.1016/j.bbr.2019.03.011.
60. Park, S.S.; Park, H.S.; Kim, C.J.; Baek, S.S.; Kim, T.W. Exercise attenuates maternal separation-induced mood disorder-like be-
haviors by enhancing mitochondrial functions and neuroplasticity in the dorsal raphe. Behav. Brain Res. 2019, 372, 112049.
https://doi.org/10.1016/j.bbr.2019.112049.
61. Zhang, L.X.; Levine, S.; Dent, G.; Zhan, Y.; Xing, G.; Okimoto, D.; Kathleen Gordon, M.; Post, R.M.; Smith, M.A. Maternal dep-
rivation increases cell death in the infant rat brain. Brain Res. Dev. Brain Res. 2002, 133, 1–11. https://doi.org/10.1016/S0926-
6410(01)00118-5.
62. Wang, Q.; Dong, X.; Wang, Y.; Liu, M.; Sun, A.; Li, N.; Lin, Y.; Geng, Z.; Jin, Y.; Li, X. Adolescent escitalopram prevents the
effects of maternal separation on depression- and anxiety-like behaviours and regulates the levels of inflammatory cytokines in
adult male mice. Int. J. Dev. Neurosci. 2017, 62, 37–45. https://doi.org/10.1016/j.ijdevneu.2017.07.007.
63. Vega-Rivera, N.M.; Fernández-Guasti, A.; Ramírez-Rodríguez, G.; Estrada-Camarena, E. Acute stress further decreases the ef-
fect of ovariectomy on immobility behavior and hippocampal cell survival in rats. Psychoneuroendocrinology 2013, 38, 1407–1417.
https://doi.org/10.1016/j.psyneuen.2012.12.008.
64. Wang, Z.; Gu, J.; Wang, X.; Xie, K.; Luan, Q.; Wan, N.; Zhang, Q.; Jiang, H.; Liu, D. Antidepressant-like activity of resveratrol
treatment in the forced swim test and tail suspension test in mice: The HPA axis, BDNF expression and phosphorylation of
ERK. Pharmacol. Biochem. Behav. 2013, 112, 104–110. https://doi.org/10.1016/j.pbb.2013.10.007.
65. Abd Al Haleem, E.N.; Ahmed, H.I.; El-Naga, R.N. Lycopene and chrysin through mitigation of neuroinflammation and oxida-
tive stress exerted antidepressant effects in clonidine-induced depression-like behavior in rats. J. Diet Suppl. 2021, 1–20.
https://doi.org/10.1080/19390211.2021.1988797.
66. Bear, T.L.; Dalziel, J.E.; Coad, J.; Roy, N.C.; Butts, C.A.; Gopal, P.K. The role of the gut microbiota in dietary interventions for
depression and anxiety. Adv. Nutr. 2020, 11, 890–907. https://doi.org/10.1093/advances/nmaa016.
67. Sonali, S.; Ray, B.; Ahmed Tousif, H.; Rathipriya, A.G.; Sunanda, T.; Mahalakshmi, A.M.; Rungratanawanich, W.; Essa, M.M.;
Qoronfleh, M.W.; Chidambaram, S.B.; et al. Mechanistic insights into the link between gut dysbiosis and major depression: An
extensive review. Cells 2022, 11, 1362. https: //doi.org/10.3390/cells11081362.
68. Kelly, J.R.; Borre, Y.; O’Brien, C.; Patterson, E.; El Aidy, S.; Deane, J.; Dinan, T.G. Transferring the blues: Depression-associated
gut microbiota induces neurobehavioural changes in the rat. J. Psychiatr. Res. 2016, 82, 109–118.
https://doi.org/10.1016/j.jpsychires.2016.07.019.
69. Rao, J.; Qiao, Y.; Xie, R.; Lin, L.; Jiang, J.; Wang, C.; Li, G. Fecal microbiota transplantation ameliorates stress-induced depression-
like behaviors associated with the inhibition of glial and NLRP3 inflammasome in rat brain. J. Psychiatr. Res. 2021, 137, 147–157.
https://doi.org/10.1016/j.jpsychires.2021.02.057.
70. Hervert-Hernández, D.; Goñi, I. Dietary polyphenols and human gut microbiota: A review. Food Rev. Int. 2011, 27, 154–169.
https://doi.org/10.1080/87559129.2010.535233.
71. Hong, M.; Zhang, R.; Liu, Y.; Wu, Z.; Weng, P. The interaction effect between tea polyphenols and intestinal microbiota: Role
in ameliorating neurological diseases. J. Food Biochem. 2022, 46, e13870. https://doi.org/10.1111/jfbc.13870.
72. Zhou, N.; Gu, X.; Zhuang, T.; Xu, Y.; Yang, L.; Zhou, M. Gut microbiota: A pivotal hub for polyphenols as antidepressants. J.
Agric. Food Chem. 2020, 68, 6007–6020. https://doi.org/10.1021/acs.jafc.0c01461.
73. Cheng, N.; Chen, S.; Liu, X.; Zhao, H.; Cao, W. Impact of schisandra chinensis bee pollen on nonalcoholic fatty liver disease and
gut microbiota in high fat diet induced obese mice. Nutrients 2019, 11, 346. https://doi.org/10.3390/nu11020346.
74. Wu, W.T.; Wang, Z.H. Effects of chrysin on intestinal inflammation and gut microbiota in lipopolysaccharides-induced mice.
FASEB J. 2019, 33, 764–768. https://doi.org/10.1096/fasebj.2019.33.1_supplement.764.8.
75. Wen, X.; Walle, T. Methylated flavonoids have greatly improved intestinal absorption and metabolic stability. Drug Metab.
Dispos. 2006, 34, 1786–1792. https://doi.org/10.1124/dmd.106.011122.
76. Paladini, A.C.; Marder, M.; Viola, H.; Wolfman, C.; Wasowski, C.; Medina, J.H. Flavonoids and the central nervous system:
From forgotten factors to potent anxiolytic compounds. J. Pharm. Pharmacol. 1999, 51, 519–526.
https://doi.org/10.1211/0022357991772790.
77. Brown, E.; Hurd, N.S.; McCall, S.; Ceremuga, T.E. Evaluation of the anxiolytic effects of chrysin, a Passiflora incarnata extract, in
the laboratory rat. AANA J. 2007, 75, 333–337.
78. Germán-Ponciano, L.J.; Puga-Olguín, A.; Rovirosa-Hernández, M.J.; Caba, M.; Meza, E.; Rodríguez-Landa, J.F. Differential ef-
fects of acute and chronic treatment with the flavonoid chrysin on anxiety-like behavior and Fos immunoreactivity in the lateral
septal nucleus in rats. Acta Pharm. 2020, 70, 387–397. https://doi.org/10.2478/acph-2020-0022.
79. Ognibene, E.; Bovicelli, P.; Adriani, W.; Saso, L.; Laviola, G. Behavioral effects of 6-bromoflavanone and 5-methoxy-6,8-dibro-
moflavanone as anxiolytic compounds. Prog. Neuropsychopharmacol. Biol. Psychiatry 2008, 32, 128–134.
https://doi.org/10.1016/j.pnpbp.2007.07.023.
80. Albert, K.; Pruessner, J.; Newhouse, P. Estradiol levels modulate brain activity and negative responses to psychosocial stress
across the menstrual cycle. Psychoneuroendocrinology 2015, 59, 14–24. https://doi.org/10.1016/j.psyneuen.2015.04.022.
Molecules 2022, 27, x FOR PEER REVIEW 18 of 18
81. Rocca, W.A.; Grossardt, B.R.; Geda, Y.E.; Gostout, B.S.; Bower, J.H.; Maraganore, D.M.; de Andrade, M.; Melton, L.J. 3rd Long-
term risk of depressive and anxiety symptoms after early bilateral oophorectomy. Menopause 2018, 25, 1275–1285.
https://doi.org/10.1097/GME.0000000000001229.
82. Giannini, A.; Caretto, M.; Genazzani, A.R.; Simoncini, T. Neuroendocrine changes during menopausal transition. Endocrines
2021, 2, 405–416. https://doi.org/10.3390/endocrines2040036.
83. Pinna, G. Allopregnanolone, the neuromodulator turned therapeutic agent: Thank you, next? Front. Endocrinol. 2020, 11, 236.
https://doi.org/10.3389/fendo.2020.00236.
84. Puga-Olguín, A.; Rodríguez-Landa, J.F.; Rovirosa-Hernández, M.J.; Germán-Ponciano, L.J.; Caba, M.; Meza, E.; Guillén-Ruiz,
G.; Olmos-Vázquez, O.J. Long-term ovariectomy increases anxiety- and despair-like behaviors associated with lower Fos im-
munoreactivity in the lateral septal nucleus in rats. Behav. Brain Res. 2019, 360, 185–195. https://doi.org/10.1016/j.bbr.2018.12.017.
85. Lovick, T.A. GABA in the female brain—oestrous cycle-related changes in GABAergic function in the periaqueductal grey mat-
ter. Pharmacol. Biochem. Behav. 2008, 90, 43–50. https://doi.org/10.1016/j.pbb.2007.12.014.
86. Rodríguez-Landa, J.F. Considerations of timing post-ovariectomy in mice and rats in studying anxiety- and depression-like
behaviors associated with surgical menopause in women. Front. Behav. Neurosci. 2022, 16, 829274.
https://doi.org/10.3389/fnbeh.2022.829274.
87. Paul, S.M.; Pinna, G.; Guidotti, A. Allopregnanolone: From molecular pathophysiology to therapeutics. A historical perspective.
Neurobiol. Stress 2020, 12, 100215. https://doi.org/10.1016/j.ynstr.2020.100215.
88. Georgieva, I.; Lepping, P.; Bozev, V.; Lickiewicz, J.; Pekara, J.; Wikman, S.; Lantta, T. Prevalence, new incidence, course, and
risk factors of PTSD, depression, anxiety, and panic disorder during the CoViD-19 pandemic in 11 countries. Healthcare 2021, 9,
664. https://doi.org/10.3390/healthcare9060664.
89. Zhukov, D.A.; Vinogradova, E.P. Neurosteroids and depression. Neurochem. J. 2021, 15, 240–246.
https://doi.org/10.1134/S1819712421030144.
90. Perez-Caballero, L.; Torres-Sanchez, S.; Romero-López-Alberca, C.; González-Saiz, F.; Mico, J.A.; Berrocoso, E. Monoaminergic
system and depression. Cell Tissue Res. 2019, 377, 107–113. https://doi.org/10.1007/s00441-018-2978-8.
91. Rana, T.; Behl, T.; Sehgal, A.; Srivastava, P.; Bungau, S. Unfolding the role of BDNF as a biomarker for treatment of depression.
J. Mol. Neurosci. 2021, 71, 2008–2021. https://doi.org/10.1007/s12031-020-01754-x.
92. Zhou, C.; Zhong, J.; Zou, B.; Fang, L.; Chen, J.; Deng, X.; Zhang, L.; Zhao, X.; Qu, Z.; Lei, Y.; et al. Meta-analyses of comparative
efficacy of antidepressant medications on peripheral BDNF concentration in patients with depression. PLoS ONE 2017, 12,
e0172270. https://doi.org/10.1371/journal.pone.0172270.
93. Sagud, M.; Nikolac Perkovic, M.; Vuksan-Cusa, B.; Maravic, A.; Svob Strac, D.; Mihaljevic Peles, A.; Zivkovic, M.; Kusevic, Z.;
Pivac, N. A prospective, longitudinal study of platelet serotonin and plasma brain-derived neurotrophic factor concentrations
in major depression: Effects of vortioxetine treatment. Psychopharmacol 2016, 233, 3259–3267. https://doi.org/10.1007/s00213-016-
4364-0.
94. Hannestad, J.; Dellagioia, N.; Bloch, M. The effect of antidepressant medication treatment on serum levels of inflammatory
cytokines: A meta-analysis. Neuropsychopharmacol 2011, 36, 2452. https://doi.org/10.1038/npp.2011.132.
95. Köhler, C.A.; Freitas, T.H.; Stubbs, B.; Maes, M.; Solmi, M.; Veronese, N.; de Andrade, N.Q.; Morris, G.; Fernandes, B.S.; Brunoni,
A.R.; et al. Peripheral alterations in cytokine and chemokine levels after antidepressant drug treatment for major depressive
disorder: Systematic review and meta-analysis. Mol. Neurobiol. 2018, 55, 4195–4206. https://doi.org/10.1007/s12035-017-0632-1.
96. Więdłocha, M.; Marcinowicz, P.; Krupa, R.; Janoska-Jaździk, M.; Janus, M.; Dębowska, W.; Mosiołek, A.; Waszkiewicz, N.; Szulc,
A. Effect of antidepressant treatment on peripheral inflammation markers-A meta-analysis. Prog. Neuro-Psychopharmacol. Biol.
Psychiatry 2018, 80, 217–226. https://doi.org/10.1016/j.pnpbp.2017.04.026.
97. Patatanian, E.; Nguyen, D.R. Brexanolone: A novel drug for the treatment of postpartum depression. J. Pharm. Pract. 2020,
0897190020979627. https://doi.org/10.1177%2F0897190020979627.
98. Patterson, R.; Krohn, H.; Richardson, E.; Kimmel, M.; Meltzer-Brody, S.A. Brexanolone treatment program at an academic med-
ical center: Patient selection, 90-day posttreatment outcomes, and lessons learned. J. Acad. Consult. Liaison Psychiatry 2022, 63,
14–22. https://doi.org/10.1016/j.jaclp.2021.08.001.




![Mechanism of action of the flavonoid chrysin potentially involved in its anxiolytic-like effects. (A) It has been confirmed that chrysin produces its anxiolytic-like effect through its action on the GABAA/benzodiazepine receptor complex producing configurational changes in the receptor and regulating the opening of the Cl − ion channel [14,15,17,24,35]; which may produce inhibitory effects in the GABAergic system associated with its anxiolytic-like effects. These effects can be blocked by specific antagonists of the GABAA receptor such as picrotoxin, bicuculline and flumazenil [14]. (B) Probably, anti-oxidant effects of chrysin could be involved in its anxiolytic-like effects. Chrysin significantly reduces ROS by inhibiting the production of NO, NT, and NOX4 [29]. These effects reduce the oxidative stress and reduces the neuronal damage. Additionally, chrysin reduces the activity of Bax, caspase-9 and caspase-3, while increasing the production of Bcl-2, thereby reducing the damage of DNA and inhibiting apoptotic processes [60,61]; which reduces the neuronal death. (C) Additionally, the anti-inflammatory effects of chrysin could contribute in its anxiolyticlike effects, considering that it may reduce the inflammatory response by inhibiting the signaling pathway NF-κB/IKK-β [27,28]. Chrysin may attenuate the expression of NF-κB that participates as transcriptional factors at nuclear level, binding to genes that induce neuro-inflammation process. Chrysin also inhibits the production of pro-inflammatory cytokines like IL-1β and IL-6, in addition to suppressing the production of proinflammatory mediators like TNF-α, PGE2 and COX-2 [27,28,32]. These effects could reduce neuro-inflammation associated with the anxiety-like behavior. ROS = reactive oxygen species; NO = nitric oxide; NT = nitrotyrosine; NOX4 = NADPH oxidase; O2¯ = superoxide; Green circles = chlorine ions; SOD = superoxide dismutase; GSH = reduced glutathione; CAT = catalase; GPx = glutathione peroxidase; Bcl-2 = anti-apoptotic protein of the subfamily Bcl-2; Bax = pro-apoptotic protein of the subfamily Bax; NF-κB = nuclear factor kappa B; IKK-β = inhibitor of nuclear factor kappa-B; TNF-α = tumor necrosis factor-α; IL-1β = interleukin-1β; IL-6 = interleukin-6; PGE2 = prostaglandins E2; COX-2 = cycloxygenase-2. (Figure was prepared by the authors).](https://www.researchgate.net/profile/Leon-German-Ponciano/publication/361003789/figure/fig1/AS:1162501056147462@1654173258979/Mechanism-of-action-of-the-flavonoid-chrysin-potentially-involved-in-its-anxiolytic-like_Q320.jpg)
![Possible mechanisms of action involved in the antidepressant-like effect of chrysin. (A) The flavonoid chrysin can modulate ERα and ERβ of membrane, which triggers the MAPK/ERK1/2 signaling pathway involved in phosphorylation and subsequently CREB activation (↑CREB), which promotes the increase of BDNF levels ( ↑ BDNF) [11,19,20,43]; which can further activate the MAPK/ERK1/2 signaling by the TrkB interaction [56]. The above-mentioned pathway also promotes an increase of TpOH expression (↑TpOH) and serotonin levels (↑Serotonin) resulting in the antidepressant-like effect [12,25]. (B) Furthermore, chrysin can decrease the pro-inflammatory cytokine levels (↓TNF-α, ↓IL-1β, ↓IL-6, ↓IFN-γ), which inhibits IDO activity (↓IDO) improving serotonergic neurotransmission and producing its antidepressant-like effect [25]. ER = estrogen receptor; MAPK = mitogen-activated-protein-kinases; CREB = cAMP response element binding; BDNF = brain derived neurotrophic factor; TrkB = tropomyosinreceptor kinase B; TpOH = tryptophan-hydroxylase; TNF-α = tumor necrosis factor-α; IL-1β = interleukin 1 beta; IL-6 = interleukin 6; IFN-γ = interferon gamma; IDO = indoleamine 2,3-dioxygenase; ERK1/2 = extracellular signal-regulated kinase 1 and 2; KYN = kynurenine. (Figure was prepared by the authors).](https://www.researchgate.net/profile/Leon-German-Ponciano/publication/361003789/figure/fig2/AS:1162501060341761@1654173259117/Possible-mechanisms-of-action-involved-in-the-antidepressant-like-effect-of-chrysin-A_Q320.jpg)