Content uploaded by Aditi Sharma
Author content
All content in this area was uploaded by Aditi Sharma on Aug 29, 2023
Content may be subject to copyright.
Vol.: (0123456789)
1 3
Eur J Plant Pathol
https://doi.org/10.1007/s10658-023-02745-5
Unveiling thebiocontrol potential ofTrichoderma
AditiSharma · BhupeshGupta· ShaliniVerma·
JoginderPal· Mukesh· Akanksha·
PraneetChauhan
Accepted: 3 August 2023
© Koninklijke Nederlandse Planteziektenkundige Vereniging 2023
Abstract Trichoderma, a well-known fungal genus
and opportunistic plant symbiont, is a quintessential
alternative to chemicals with great potential to mini-
mize disease incidence. The mycoparasitic ability
along with antibiosis and induction of host immunity
are the main mechanisms of biocontrol by this fun-
gus. Fungi belonging to genus Trichoderma have been
identified as potential biocontrol agents due to major-
ity of isolated antifungal bioactive compounds. This
review summarizes the biological control activity
exerted byTrichodermaspp. against plant pathogenic
fungi, bacteria, viruses, nematodes and insect pests.
In addition, the research on formulations advocates
that encapsulation could be a promising tool for
increasing efficacy and durability of these fungi
under field conditions. Further, advances in different
areas of science and technology would strengthen the
future research on Trichoderma-based products for its
efficient use in agriculture.
Keywords Trichoderma· Biological control·
Bioactive compounds· Parasitism· Formulation
Introduction
Plant pathogens pose a major threat to food security,
and their management has become a big challenge in
modern times. In the name of intensive agriculture,
the indiscriminate use of chemicals has resulted in
negative outcomes in the past few years. Many exam-
ples from previous research into the heavy reliance of
agriculture on synthetic pesticides and fertilizers have
shown negative impacts of these molecules on human
and plant health, the environment, and on the ecosys-
tem (Tilman, 1998 and 1999; Nicolopoulos-Stamati
etal., 2016). Moreover, plant pathogens have become
resistant to several chemicals, and these molecules
also affect non-targeted organisms (Padovani et al.,
2004; Sharma etal., 2019; Topping etal., 2020). The
biological control of pathogens by antagonistic liv-
ing microorganisms is a promising approach for the
management of plant diseases and pests (Ab Rahman
etal., 2018; Ons etal., 2020). There are many bacteria
A.Sharma(*)
College ofHorticulture andForestry, Thunag- Mandi,
Dr. Y. S. Parmar University ofHorticulture andForestry,
Nauni, Solan173230, HimachalPradesh, India
e-mail: aditi.bhardwaj650@gmail.com
B.Gupta· S.Verma· Mukesh· Akanksha
Department ofPlant Pathology, Dr. Y.S. Parmar University
ofHorticulture andForestry, Nauni, Solan173230,
HimachalPradesh, India
J.Pal
Department ofPlant Pathology, CSK Himachal
Pradesh Krishi Vishwavidyalaya, Palampur,
HimachalPradesh176062, India
P.Chauhan(*)
Department ofPlant Pathology, Dr Khem Singh Gill Akal
College ofAgriculture, Eternal University, Barusahib,
SirmourHimachalPradesh173101, India
e-mail: chauhanpraneet78@gmail.com
Eur J Plant Pathol
1 3
Vol:. (1234567890)
(such as Pseudomonas, Bacillus, Paenibacillus, and
Streptomyces), fungi (such as Trichoderma, Glomus,
Beauveria, endophytic fungi) and viruses that are
being deployed for the control of various pests and
pathogens (Latz etal., 2018; Raymaekers etal., 2020;
Sala etal., 2019). Among fungal biocontrol agents the
Trichoderma genus is the most extensively studied and
exploited.
Weindling in 1932 identified the genus Tricho-
derma as having biocontrol potential against plant
pathogens (Weindling, 1932 and 1934). These fungi
are ubiquitous, colonize above and belowground
plant organs, grow as endophytes, and are among
the predominant components of soil mycoflora. The
ability of Trichoderma to sense, invade and attack
pathogenic fungi has been the impelling cause for its
commercial success as biopesticides (Hermosa etal.,
2012; Verma etal., 2007). The use of Trichoderma
harzianum against Sclerotium rolfsii in 1972 was the
first successful example of biocontrol efficacy under
field conditions (Wells et al., 1972). Trichoderma,
a genus of familyHypocreaceae, are asexual fungi
that generally reproduce via chlamydospores, can
survive at different pH ranges, and have an optimum
temperature range of 25–35°C (Shah & Afiya, 2019).
These antagonistic fungi can be most frequently iso-
lated from soils, and temperate and tropical zones
contain approximately 101 –103 culturable spores of
Trichoderma per gram of soil (Vinale etal., 2008).
Generally, the morphological taxonomy of Tricho-
derma is based on fungal hyphae, spore size, shape,
colony color and appearance of mycelium on specific
culture media. However, molecular techniques are
more precise and reliable methods to discover diverse
Trichoderma species (Atanasova et al., 2013; Bis-
settet al., 2003; Hewedy etal., 2020; Kamala etal.,
2015). Due to the diverse use of these fungi, quick
and accurate identification is necessary. Researchers
and taxonomists around the globe have developed a
series of identification tools that make Trichoderma-
based studies more accessible (Dou etal., 2020; Dru-
zhinina etal., 2005; Hebert etal., 2003; Raja etal.,
2017). The genus Trichodermacomprises 495 iden-
tified species based on molecular phylogeny, and
they show a variety of biological activities (Har-
manet al., 2004; Druzhinina etal., 2006; Bissettet al.,
2015; Qin & Zhuang, 2016; Index Fungorum, 2022).
There are several well known species of Trichoderma
such as T. harzianum, T. hamatum, T. asperellum,
T. viride, T. koningii, T. pseudokoningii, T. afro-
harzianum and T. cyanodichotomus that have been
identified as promising biocontrol agents (del Car-
men etal., 2021; Mukherjee etal., 2013). Genomic
studies have facilitated a more advanced understand-
ing of the phylogenetic relationships of these fungi,
together with furthering investigations on sexual
crossings, and their role in ecosystems (Zhou etal.,
2020). The capacity of Trichoderma spp. to produce
different enzymes, antibiotics, to induce resistance in
plants, and to act as parasites of different pathogens,
make these fungi the most studied biopesticide and
biofertilizer (Solanki etal., 2011; Waghunde etal.,
2016). Furthermore, the development of formula-
tions of Trichoderma spp. ensures the protection of
the active ingredient under extreme pH, humidity,
chemicals and UV radiation conditions, giving the
biocontrol fungi a competitive advantage over major
pathogenic microbes. Formulations of Trichoderma
spp have mostly adopted solid or liquid fermentation
technologies together with various organic and inor-
ganic carriers (Sanjeev etal., 2014). Recent advances
in research have shown the use of novel methods for
the development of formulations, which can retain
conidial viability and work well under field con-
ditions (Locatelli et al., 2018; Swain et al., 2021;
Yobo etal., 2019).
Overview ofthegenus Trichoderma
andmechanism ofbiocontrol
The genus Trichoderma, belonging to the fam-
ily Hypocreaceae, is present in all kinds of soils
and can be cultured on artificial media. Persoon, in
1794, first proposed Trichoderma as a genus on the
basis of material collected in Germany. Later, in
1865, the Tulasne brothers in France illustrated the
link between T. viride and Hypocrea rufa (Persoon,
1794; Tulasne & Tulasne, 1865). Similarly, Wein-
dling was a pioneer in proposing Trichoderma as a
fungus that parasitizes other fungi, and demonstrated
its biocontrol potential in 1932 (Mukhopadhyay &
Kumar, 2020). These fungi form filamentous hyphae
with abundant conidiophores. The asexual conidia
are hyaline, elliptical in shape, and form white to yel-
low mycelium and green conidia on culture media
when completely mature (Atanasova et al., 2013;
Jaklitsch, 2009). In agriculture different Trichoderma
Eur J Plant Pathol
1 3
Vol.: (0123456789)
spp, such as T. atrobrunneum, T. afroharzianum, T.
guizhouense, T. harzianum, T. simmonsii, T. asperel-
lum, T. asperelloides, T. atroviride, T. hamatum, T.
gamsii, T. koningii, T. koningiopsis, T. viridescens, T.
citrinoviride, T. longibrachiatum, T. pseudokoningii,
T. saturnisporum, T. cyanodichotomus, T. spirale and
T. polysporum, have been deployed to control various
diseases and insect pests (Mastouri etal., 2012; Rush
etal., 2021; Sood etal., 2020; Woo etal., 2014). The
biocontrol and symbiotic properties of Trichoderma
are the most important factors that determine its
widespread application in agriculture. According to
Rush etal. (2021), of all the fungal biocontrol agents,
50–60% belong to the genus Trichoderma. A recent
review by Tyśkiewicz etal. (2022), states that approx-
imately 77 commercial Trichoderma-based biofun-
gicides are available and approved by the European
Union (Thambugala etal., 2020). The vast majority
of Trichoderma species are not associated with their
sexual state and therefore differentiation of species
within the genus becomes very difficult. However,
enormous advances have been made in the taxonomy
of Trichoderma based on DNA Barcoding and Multi-
locus Identification System (MIST) (Cai etal., 2022;
Tang etal., 2022).
The powerful biocontrol machinery of Tricho-
dermaspp. is the driving unit for its use as prophy-
lactic and curative treatments against plant pathogens,
particularly when chemical methods become inef-
fective or economically unfeasible. Trichoderma is
known to exhibit several direct and indirect biocontrol
mechanisms against different pathogens. The myco-
parasitic effect of Trichoderma involves the sensing
of prey, chemotaxis, adhesion to host surfaces, and
physical attack through coiling and intense branching
of mycelium around the pathogen hyphae (Mukherjee
etal., 2012; López- Bucio etal., 2015; Guler etal.,
2016; Redda et al., 2018). They are also known to
produce low-molecular-weight volatile or nonvola-
tile antibiotics or secondary metabolites that restrict
the growth of pathogens. The fungus releases several
glucanases, chitobioses, chitinase enzymes, antibiot-
ics like viridin, gliotoxin and peptaibols (Dou etal.,
2020). These biocontrol fungi are also considered one
of the richest sources of peptaibols, and over 80% of
peptaibiotics entered in the database is synthesized by
this genus (Neumann etal., 2015). In addition, T. har-
zianum is known to produce harzianic acid that shows
antifungal activity, stimulation of plant growth and
chelating properties (Vinale etal., 2009). It has been
well established that Trichoderma species compete
very well against pathogens for nutrients and space in
the soil and rhizosphere (Solanki etal., 2011). Fungi
belonging to this genus are also well known to indi-
rectly affect pathogenic microbes by inducing local or
systemic defense mechanisms in plants. Trichoderma
activates two types of induced resistance in plants,
i.e. systemic acquired resistance (SAR) and induced
systemic resistance (ISR) (Baiyee etal., 2019; Etim
& Onah, 2022; Shoresh etal., 2010). Besides its bio-
efficacy against pathogens, it has been suggested by
many researchers that the presence of Trichoderma
in rhizosphere leads to increased tolerance towards
abiotic stresses (Ghorbanpour et al., 2018; Tripathi
et al., 2021). Additionally, Trichoderma spp. also
possess plant growth promoting properties (Halifu
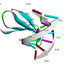
etal., 2019). Figure1 depicts the multifaceted role of
Trichoderma.
Antifungal properties ofTrichoderma
Pathogenic fungi cause serious problems to the pro-
duction of agricultural commodities both at pre and
post harvest stages. These fungi exhibit huge vari-
ability in terms of their mode of infection, reproduc-
tion, spore formation and nutritional strategy (Agrios,
2005). They are regarded as one of the dominant
causes of plant diseases. A plethora of strategies to
colonize and invade plants have been developed by
these microbes. They are generally classified into
four different phyla namely, Chitridiomycota, Oomy-
cota, Ascomycota and Basidiomycota. Among these
phyla Synchytrium, Phytophthora, Pythium, Magna-
porthe, Erysiphe, Cladosporium, Botrytis, Fusarium,
Colletotrichum, Rhizoctonia, Puccinia and Ustilago
are some important genera, causing huge economic
losses to crops every year (Doehlemann etal., 2017).
Trichoderma is one of the most widely applied bio-
control agents used in plant disease management. Dif-
ferent species of Trichoderma multiply rapidly, and
advantageously inhibit the growth of fungi by hyper-
parasitic effect (Sreenivasaprasad & Manibhushan-
rao, 1990). There are numerous examples of hyper-
parasitism of Rhizoctonia solani by different species
of Trichoderma by coiling the mycelium of the path-
ogenic fungi (Elad etal., 1983; Inayati etal., 2020;
Naeimi etal., 2010). Recently, hyperparasitism by this
Eur J Plant Pathol
1 3
Vol:. (1234567890)
fungi has also been reported against Epichloespecies
that causes “choke disease” of grasses (Górzyńska
et al. 2018; Węgrzyn & Górzyńska, 2019). It has
been reported that when Trichoderma is cocultured
with Rhizoctonia, hyperparasitic functional genes and
antibiosis genes are upregulated (Halifu etal., 2020).
Rice sheath blight, caused by Rhizoctonia solani,
is one of the major constraints in the production of
Oryza sativa L. (rice) and can be effectively managed
by different species of Trichoderma (Safari Motlagh
etal., 2022). According to Xu etal. (2022), intercrop-
ping of maize and soybean significantly increased
the density and composition of Trichoderma and
antagonized pathogenic Fusarium species in the
rhizosphere. It has also been documented that pro-
tective treatment with T. asperelloidesenhances the
expression of defense genes (viz., PR-1, PR-2, PR-3,
and CHS) resulting in a lower incidence of root rot
in Solanum lycopersicumL. (Heflish etal., 2021). In
addition to this, Trichoderma produces several vola-
tile or nonvolatile compounds that inhibit pathogen
growth. The antibiosis and mycoparasitic activity
of these biocontrol fungi has been shown to reduce
the growth of Rhizoctonia solani, which causes seed
rot in Gossypium L. (Gajera et al., 2020). Cultural
extracts of T. asperellum andMetarhizium anisopliae
inhibit conidial germination, and exhibit chitinolytic
activity against Leveillula taurica (Guigón López
etal., 2019). Trichoderma longibrachiatum produces
several secondary metabolites that inhibit fungal
pathogens such as Colletotrichumlagrnarium, Bot-
rytis cinerea, and Fusarium oxysporum (Du et al.,
2020). Hydrolytic activity of enzymes produced by
T. harzianum, T. guizhouense, T. atroviride and T.
Fig. 1 Multifaceted role of Trichoderma
Eur J Plant Pathol
1 3
Vol.: (0123456789)
koningiopsis also reduced the incidence of Penicil-
lium digitatum Sacc.,Alternaria alternata(Fr.) Keissl
andColletotrichum gloeosporioides (Penz.) Penz. &
Sacc. (Ferreira et al., 2020). Volatile organic com-
pounds of T. asperellum,T. harzianum, andT. atrovir-
ide reduced the growth of Plasmopara viticola (P. vit-
icola,Berk. & M. A. Curtis; Berl. & De Toni), which
causes grapevine downy mildew (Lazazzara et al.,
2021). A recent study reports the mycoparasitic action
of T. asperellum, T. harzianum, T. hamatum, T. vir-
ideand T. longibrachiatum against Biopolaris oryzae,
Rhizoctonia solani, Fusarium semitectumandCurvu-
laria oryzae (Klaram etal., 2022). Further, xylanase
secreted byT. asperellum stimulates systemic resist-
ance in poplar (Populus L.) againstAlternaria alter-
nata, Rhizoctonia solani, and Fusarium oxysporum
(Guo etal., 2021a). Biopriming with Trichoderma is
also an adaptive strategy to enhance the plant defense
against pathogenic microbes. A recent report docu-
mented that plants primed with T. harzianumshowed
higher activity of resistance enzymes (such as super-
oxide dismutase, catalase, peroxide, phenylalanine
ammonia lyase and polyphenol oxide) compared
to plants inoculated with Fusarium oxysporum f.
sp. lycopersici (Zehra etal., 2023). Biocontrol with
T. asperellum induced resistance against Puccinia
striiformis f. sp. tritici, causal agent of stripe rust of
wheat (genus Triticum; L.), as reported by Esmail
et al. (2023). Table 1 depicts some research on the
antifungal effects of different Trichoderma species
against pathogenic fungi in agriculture.
Antibacterial properties ofTrichoderma
Plant pathogenic bacteria cause serious disease out-
breaks in many economically important crops with
severe consequences on food production and secu-
rity. Infected plants exhibit different types of damag-
ing symptoms such as blight, canker, galls and over-
growth, wilts, leaf spots, specks, scab and soft rots
(Agrios, 2005). Phytopathogenic bacteria contain
various virulence factors that enable them to infect
and colonize host tissues successfully. Due to wide
host ranges and the development of antibiotic resist-
ance, their management has become a challenging
task. Thus, many methods are now being adopted
for developing effective strategy to control these
pathogenic prokaryotes. One of the best strategies is
biocontrol of bacterial pathogens with different antago-
nistic microbes (Sharma etal., 2017). The potential of
Trichoderma in controlling the growth of pathogenic
bacteria has been demonstrated by different research-
ers. Different species of Trichoderma have shown
induced resistance against Xanthomonas euvesicato-
riain tomato plants (Fontenelle etal., 2011). Tricho-
derma asperellum produces siderophores, indole-
3-acetic acid, hydrogen cyanide, and salicylic acid.
These secondary metabolites are known to inhibit the
growth of Ralstonia solanacearum that causes wilt
in potato (Solanum tuberosum L.) (Mohamed et al.,
2020). Bacterial pathogens utilize different patho-
genicity factors such as secretion systems (type I, II,
III, IV), quorum sensing (QS), enzymes, toxins, hor-
mones, polysaccharides, proteinases and siderophores
that contribute to disease development in host plants
(Siphathele et al., 2018). Thus, bioactive compounds
secreted by the biocontrol microbes must target these
virulence factors for effective disease control. Bio-
active compounds produced by T. harzianum have
shown high efficacy in inhibiting bacterial patho-
gens such as Xanthomonas campestris, Clavibacter
michiganensis, Escherichia coli, and Pseudomonas
aeruginosa (Anwar & Iqbal, 2017). Biofilm protects
bacteria against adverse environmental conditions and
the quorum sensing mechanism has been targeted by
researchers for management of bacterial pathogens. A
secondary metabolite produced by T. atroviride was
reported to interfere with the gene pathways involved
in biofilm formation of Xanthomonas campes-
trispv.campestris that causes black-rot disease in cru-
cifers (Papaianni etal., 2020). Secondary metabolites
of Trichoderma koningiopsisalso inhibited the growth
of Erwinia mallotivora, the causal agent of papaya
(Carica papaya L.) dieback disease (Tamizi et al.,
2022). Antibacterial enzymes produced by T. harzi-
anumhave shown inhibitory activity against a highly
virulent bacterium, Erwinia amylovora (Smirnova
et al., 2017). Similar activity of Trichoderma spp.
against Erwinia carotovora has been reported by
Sulaiman et al. (2020). Secondary metabolites of T.
koningiopsis have also been shown to have biocontrol
activity against Erwinia mallotivora that causes die-
back in papaya (Tamizi etal., 2022). The potential of
Trichoderma to control other important bacteria such
as Ralstonia solanacearum causing wilt was demon-
strated by different workers. Crude extract contain-
ing secondary metabolites of this fungus when tested
Eur J Plant Pathol
1 3
Vol:. (1234567890)
Table 1 Antifungal action of different Trichoderma species
Trichoderma species Fungal Pathogens Disease Mechanism Reference
T. asperelloidesandT. afroharzianum Erysiphe necator Grapevine powdery mildew Enzymatic distortion of conidial
structure
Sawant etal., 2017
T. longibrachiatum, T. afroharzi-
anum, T. koningiop, T. citrinovi,
T. rossicum, T. gamsii
Fusarium oxysporum, Rhizoctonia
solani and Botrytis cinerea Wilt, blight and rot Mycoparasitism Redda etal., 2018
Trichoderma viride Phytophthora infestans Late blight Induction of resistance Purwantisari etal., 2018
Trichoderma harzianum,
Trichoderma longibranchia-
tum, Trichoderma yunnanense,
Trichoderma asperellum
Colletotrichum gloeospori-
oidesandPhytophthora capsici Tomato anthracnose, Blight and
fruit rot of chilli
Production of cellulases and
chitinases
De la Cruz-Quiroz etal., 2018
Trichoderma harzianum Phytophthora cactorumandPhy-
tophthoraspp.
Collar rot of pear Mycoparasitism Sanchez etal., 2019
Trichoderma asperellum Corynespora cassiicolaandCur-
vulariaaeria Leaf spot of lettuce Induced defense response Baiyee etal., 2019
Trichoderma longibrachiatum Colletotrichum spp. Botrytis
cinerea, and Fusarium oxyspo-
rum
Anthracnose, grey mold rot and
wilt
Secondary metabolite production Du etal., 2020
Trichoderma harzianum,T.
guizhouense,T. atrovirideandT.
koningiopsis
Penicillium digitatum,Alternaria
alternataandColletotrichum
gloeosporioides
Green mould, leaf spot, anthrac-
nose of citrus
Hydrolytic activity of enzymes Ferreira etal., 2020
Trichodermalongibrachiatum, T.
harzianum, T. pleuroti Sclerotinia sclerotiorum,Scle-
rotium rolfsii andFusarium
oxysporum, Macrophomina
phaseolina
Rot, Blight, Wilt and Charcoal
Rot
Mycoparasitism and volatile
organic compounds
Rajani etal., 2021
T. gamsii,T. viridarium,T. hama-
tum,T. olivascens,T. virens,T.
paraviridescens,T. linzhiense,T.
hirsutum,T. samuelsii, andT.
harzianum
Phytophthora cinnamomi Oak Decline Mycoparasitism Ruiz-Gómez & Miguel-Rojas,
2021
Trichoderma asperellum,T. harzi-
anum, andT. atroviride Plasmopara viticola Grapevine downy mildew Volatile organic compounds Lazazzara etal., 2021
Trichoderma erinaceum Pythium ultimum Damping off and root rot Secondary metabolites Siebatcheu etal., 2023
T. asperellum, T. harzianum, T.
hamatum, T. virideand T. longi-
brachiatum
Biopolaris oryzae, Rhizoctonia
solani, Fusarium semitec-
tumandCurvularia oryzae
Brown spot, sheath blight, leaf
blight
Mycoparasitism Klaram etal., 2022
Eur J Plant Pathol
1 3
Vol.: (0123456789)
in vitro and in planta for the management ofbacte-
rial wilt resulted in effective control (Yan & Khan,
2021). Zhang etal. (2022) reported that trichokonins
A, peptaibols produced byTrichoderma longibrachia-
tum SMF2 inhibit the growth of Xanthomonas ory-
zae pv. oryzae with a minimum inhibitory concentra-
tion of 54μg/mL. Trichokonin A resulted in damage
to bacterial cell membranes and loss of permeability.
Similarly, another peptaibol analog (named Trichogin
GA IV) produced by Trichoderma longibrachiatum
has shown antibacterial action against Xanthomonas
campestris pv. campestris that causes black rot of cru-
cifers (Caracciolo etal., 2023). There are reports that
suggest a vital role in defense induction by this fungus
against bacterial blight causing pathogens. Activa-
tion of reactive oxygen species and induction of PR
proteins by T. asperellum was documented against
Xanthomonas oryzae pv. oryzae (Singh etal., 2019).
Recently, Islam etal. (2023) advocated that four strains
of Trichoderma (viz., T. paraviridescens, T. para-
viridescens, T. erinaceumand T. asperellum)induced
resistance against X. oryzaepv.oryzae by upregulating
core defense-related genes such as OsPR1,OsPR10,O
sWR KY4 5, OsWRKY62, OsWRKY71, OsHI-LOX,
and OsACS2 after 24 h of inoculation. The attain-
ment of sustainable bacterial disease management is
extremely difficult and continuously increasing the
amount of dataon the biocontrol potential of Tricho-
derma can play a vital role in generating effective strat-
egies for bacterial disease management. In a recent
study on transgenic plants it was advocated that a gene
encoding a sphingomyelinase, identified in T. harzi-
anum, generated disease resistance to Pseudomonas
syringae pv. tabaci and tolerance to Xylella fastidi-
osa. This gene was expressed in all transgenic tobacco
lines that were tested and can be utilized in molecular
breeding to achieve resistance against pathogens (Ber-
bert etal., 2022). Thus, these findings further strength-
ened the fact of using these potential antagonistic fungi
against bacterial plant pathogens (Table2).
Antiviral properties ofTrichoderma
Plant viruses comprise one of the most important
groups of plant pathogens, sometimes causing up to
100% yield loss due to systemic infection. In the present
scenario, nearly 47% of the emerging and re-emerg-
ing plant pathogens are viruses (Saikia et al., 2022).
Viruses are notorious mesobiotic pathogens that are
responsible for huge economic losses in many crops
due to their ability to sequester and reprogram the host
cellular machinery for their replication. The manage-
ment of viruses is very difficult and indirect due to their
transmission by biotic agents like many insects, fungi
and nematodes. The exploitation of antagonistic poten-
tial, resistance induction and plant-growth-promoting
activities of Trichoderma spp. is a well-investigated
area in plant protection. Therefore, many researchers
have exploited Trichoderma spp. for the management of
viral pathogens through induction of host resistance.
Tobacco mosaic virus (TMV) is a single-stranded
RNA virus that infects many important plant species,
essentially tobacco and tomato plants (Ge etal., 2012;
Scholthof etal., 2011). An endophytic fungal strain,
T. koningii produced 6-pentyl-α-pyrone that induced
systemic resistance in Nicotiana tabacumcv. White
Burley against TMV (Taha et al., 2021). In addi-
tion to this, nigirpexin (azaphilone derivatives), a
compound obtained from one strain of T. afroharzi-
anumhas also shown antiviral activity against TMV
(Xie etal., 2022). Abdelkhalek etal. (2022) reported
the protective activity of T. hamatumagainst tobacco
mosaic virus. Trichoderma inoculated plants showed
significant increases in reactive oxygen species scav-
enging enzymes thereby inducing defence in plants.
Cucumber mosaic virus (CMV) is one of the most
widespread and harmful plant viruses, infecting more
than 1200 plant species worldwide and it is efficiently
transmitted by more than 80 aphid species (Dubey
& Singh, 2010). A strain of T. asperellum induced
resistance against yellow strain ofCucumber mosaic
virus in Arabidopsis plants through salicylic acid
(SA) signaling cascade (Elsharkawy etal., 2013).The
molecular mechanisms underlying T. asperel-
lummediated resistance were revealed as the upregu-
lation of resistance-related genes such as pr1, pal1,
etr1, sod, rip and lox1. This biocontrol fungus also
increased the hydrogen peroxide (H2O2) content in
plants, thereby inducing systemic resistance against
CMV (Tamandegani et al., 2021). T. harzianum in
combination with other biocontrol fungi has proven
to activate resistance against Maize chlorotic mottle
virus(MCMV) andSugarcane mosaic virus(SCMV)
that are generally transmitted by thrips and aphids.
Both of these viruses pose serious threats to cereal
production and result in a severe viral syndrome
known as maize lethal necrosis (MLN)(Kiarie etal.,
Eur J Plant Pathol
1 3
Vol:. (1234567890)
Table 2 Antimicrobial and insecticidal properties of different Trichoderma species
Antibacterial action of Trichoderma
Trichoderma species Pathogens/Pest Disease /Order Mechanism Reference
Trichoderma asperellum Ralstonia solanacearum Wilt Induction of plant resistance Konappa etal., 2018
Trichoderma viride Xanthomonas citri Canker Enzymatic activity Jatav etal., 2018
Trichoderma asperellum Xanthomonas
oryzaepv.oryzae Bacterial blight
of rice
Oxidative burst-mediated
defense
Singh etal., 2019
Trichoderma atroviride Xanthomonas campes-
trispv.campestris Black rot Antibiofilm activity of
metabolite
Papaianni etal., 2020
Trichoderma harzi-
anum,Trichoderma
virideandTrichoderma
virens
Pectobacterium carotovo-
rumsubsp.carotovorum Soft rot Growth inhibition Abd-El-Khair etal., 2021
Trichodermaharzianum,T.
virensandT. koningii Ralstonia solanacearum Wilt Secondary metabolites Guo etal., 2021b
Trichoderma harzianum Clavibacter michiganen-
sissubsp.michiganensis Tomato wilt Antagonistic activity Abo-Elyousr etal., 2022
Trichoderma koningiopsis Erwinia mallotivora Papaya Dieback Secondary metabolites Tamizi etal., 2022
Antiviral action of Trichoderma
Trichoderma sp. Zucchini yellow mosaic
virus (ZYMV)
Squash Induction of systemic
resistance
Abdel-Shafi etal., 2013
Trichoderma harzianum Cucumber mosaic
virus(CMV) Solanum lycoper-
sicum
var.cerasiforme
Induced systemic resistance Vitti etal., 2016
Trichoderma albolutescens Pepper mottle virus (PMV) Tobacco and hot
pepper
Antiviral Trichothecenes Ryu etal., 2017
Trichoderma koningii Tobacco mosaic
virus(TMV)
Tobacco Increased the activities
of pathogenesis-related
enzymes
Taha etal., 2021
Trichoderma asperellum,
Trichoderma longibra-
chiatum, and Trichoderma
asperlloides
Tomato yellow leaf curl
virus (TYLCV)
Tomato Resistance Al Abedy etal., 2021
Trichoderma harzianum Papaya ringspot virus
(PRSV) Cucumeropsis
mannii Resistance induction Etim & Onah, 2022
Nematicidal action of Trichoderma
Trichoderma longibrachiatum Heterodera avenae Molya disease of
wheat
Direct parasitic and lethal
effect on eggs and
resistance induction
Zhang etal., 2017
Trichoderma atroviridae Globodera rostochiensis Golden potato
cyst
Chitinolytic activity Abbasi etal., 2017
Trichoderma atroviride Meloidogyne javanica Root-knot Induction of plant resistance Medeiros etal., 2017
Trichoderma harzianum Meloidogyne incognita Root-knot Induction of plant resistance Singh etal., 2017
Trichoderma asperellum,T.
atroviride,Trichodermasp. Meloidogyne javanica Gall Reduction of gal egg
mass and eggs
Kiriga etal., 2018
Trichoderma viride Meloidogyne javanica Root-knot Direct antagonism, lytic
enzymes production and
induction of defense
responses
Hajji-Hedfi etal.,2021
T. harzianum Meloidogyne incognita Root-knot Secondary metabolites
and defense-related
enzyme activity
Yan etal., 2021
Eur J Plant Pathol
1 3
Vol.: (0123456789)
2020). Whitefly transmitted pepper leaf curl virus
(PeLCV) is also a cosmopolitan plant virus that ham-
pers a wide range of crops. The suppressive effects
of T. polysporum, T. atroviridae, T. harzianum and
their consortia were analyzed against PeLCV by
Rochal etal. (2021). In this study, authors reported
Trichoderma-induced innate host immunity prim-
ing and stress tolerance in plants against Pepper leaf
curl virus. The role ofTrichoderma in triggering the
plant immunity has been studied in isolation both at
the local and systemic level. In this context, various
signature molecules fromTrichoderma,together with
their roles in activating the plant immune responses,
have been identified (Salwan et al., 2022). Table 2
depicts the research findings of different research-
ers advocating for the biocontrol potential of Tricho-
dermaagainst viruses.
Nematicidal properties ofTrichoderma
Nematodes are ubiquitous, free-living microscopic
entities that result in 12—25% annual losses in sev-
eral economically important crops (Agrios, 2005;
Bernard etal., 2017; Jones etal., 2013; Nicol etal.,
2011; Seinhorst, 1970). There are more than 4100
plant parasitic nematodes, of which the root-knot
nematodes and cyst nematodes have the most detri-
mental effects on crops (Decraemer & Hunt, 2006;
Jones et al., 2013; Mai & Abawi, 1987). Efficient
eco-friendly methods are necessary for the manage-
ment of nematodes, as nematicides lead to environ-
mental hazards and resistance development (Chen
etal., 2020). Therefore, researchers are focusing on
the use of biocontrol methods against nematodes to
reduce the losses to agricultural comodities (Moosavi
& Zare, 2020).
Along with other antagonistic fungi, significant
research has been carried out on the use of various
Trichoderma spp. for controlling plant-parasitic nem-
atodes (Sharon etal., 2011). The direct antagonistic
mechanism of these fungi against nematodes involves
parasitism of nematode eggs and juveniles, conidial
attachment on the nematode surfaces, hyphae coiling
around second generation juveniles, and enzymatic
lysis of eggs (Sahebani & Hadavi, 2008). Tricho-
derma harzianum possesses strong egg-parasitic
ability, and the chitinolytic enzymes produced by the
Table 2 (continued)
Trichoderma asperellumand
Trichoderma harzianum Pratylenchus brachyurus Root lesion Non-volatile metabolites de Oliveira etal., 2021
T. citrinoviride, T. ghanense,
T. harzianum, T. koningi-
opsis, T. simmonsii, and
T. virens
Meloidogyne javanica and
M. incognita Root-knot Sesquiterpene and pol-
yketide-like metabolites
Moo-Koh etal., 2022
Insecticidal action of Trichoderma
T. longibrachiatum Leucinodes orbonalis Lepidoptera Parasitism Ghosh & Pal, 2016
T. longibrachiatum Bemisia tabaci Hemiptera Parasitism Anwar etal., 2016
T. viride Corcyra cephalonica Lepidoptera Production of antifeedant
compounds
Vijayakumar and Alagar,
2017
T. harzianumT. atroviride T.
citrinoviride Xylotrechus arvicola Coleoptera Parasitism Rodríguez-González
etal., 2017
T. viride T. harzianum Odontotermes formosanus Blattodea Repellent activity Xiong etal., 2018
Trichoderma sp Aphis gossypii Hemiptera Secondary metabolites
toxicity
Nawaz etal., 2020
Trichoderma sp. Locusta migratoria Orthoptera Secondary metabolites
toxicity
Laib etal., 2020
Trichoderma koningiopsis Diatraea saccharalis Lepidoptera Enhanced insecticidal
activity of Beauveria
bassiana
Mejía etal., 2021
Trichoderma chlorosporum Philaenus spumarius Hemiptera Secondary metabolites
toxicity
Ganassi etal., 2022
Eur J Plant Pathol
1 3
Vol:. (1234567890)
fungi results in lysis of nematode eggs (Szabó etal.,
2012). The reduction in nematode infestation by chi-
tinase and protease production is the most important
attribute in Trichoderma biocontrol (Sharon et al.,
2011). Chitinolytic activity of T. asperelloides has
been recorded against M. incognita (Sayed et al.,
2019). In addition to enzyme activity, a significant
antibiosis effect of different secondary metabolites of
T. harzianumisolates has been reported that result in
strong mortality of nematode juveniles (Babu etal.,
2019). Nematicidal metabolites of this fungi have
been isolated and characterized by different workers,
and are categorized as acetic acid, gliotoxin, trichor-
zianine, viridin trichodermin and cyclonerodiol
(Li etal., 2007; Zhou et al., 2018; Shi etal., 2020).
Several compounds classified in the koninginins
group, isolated from solid fermentation products
ofT. neokongii, also showed nematicidal properties
(Lang etal., 2015; Zhou etal., 2014). Similarly, dif-
ferent nematicidal compounds from diverse species
of Trichoderma are being harnessed to develop for-
mulations against parasitic nematodes such asMeloi-
dogyne incognita,M. javanica,M. hapla, Globodera
pallida,Heterodera avenae,andPratylenchus brach-
yurus (Braithwaite et al., 2016; Tariq-Javeed etal.,
2021). Sesquiterpene and polyketide-like metabo-
lites, produced by strains of T. harzianum, T. kon-
ingiopsis, T. ghanense, and T. virens, have shown
51–100% mortality of second-stage juveniles of M.
javanica and M. incognita (Moo-Koh etal., 2022).
Volatile organic compounds (VOCs) of Trichoderma
inhibit populations of Panagrellus redivivus, Cae-
norhabditis elegans, and Bursaphelenchus xylophi-
lus (Yang et al., 2012). Besides these mechanisms,
the role of induced systemic resistance (ISR) has
been well documented in recent years (Goverse &
Smant, 2014). Current research is focusing on uti-
lizing jasmonic acid regulated defences by Tricho-
derma against nematodes (Martínez-Medina et al.,
2017). There are reports that support the mecha-
nism of induced systemic resistance by Trichoderma
against M. incognita (Pocurull et al., 2020). Nowa-
days, different species of Trichoderma are being
deployed that show lethal effects against nematodes
(Elgorban etal., 2014; Braithwaite etal., 2016; Fan
etal., 2020).Trichoderma harzianumandT. asperel-
lum have shown biocontrol potential against juve-
niles of M. javanica in banana (Genus Musa L.)
(Almeida etal., 2022). There are various examples of
the nematicidal effect of different Trichoderma spp.
and their mode of action (Table2).
Insecticidal properties ofTrichoderma
Insects are considered important pests in agricultural
and horticultural production systems, causing huge
losses to crops during both production and storage.
According to recent estimates, insect pests result in
18- 25% average losses in agricultural productivity
worldwide. These losses are generally more common
in food-deficit regions with fast-growing populations,
and may also be due to emerging or re-emerging pests
(Srivastava & Subramanian, 2016). However, indis-
criminate use of pesticides for controlling insect pests
has led to pollution of soil, water, food sources, as
well as poisoning of nontarget insects and the devel-
opment of resistance. To obviate these harmful effects
and obtain higher yields, biocontrol of insect pests
using specific antagonistic microbes is an effective
alternate strategy (Sindhu etal., 2017). The manage-
ment of these insect pests is very difficult, and several
workers are concentrating on using ecofriendly meth-
ods to manage them (Litwin etal., 2020; Manivel &
Rajkumar, 2018). There are many reports that have
determined the insecticidal potential of Trichoderma
in various crops (Battaglia et al., 2013; Contreras-
Cornejo et al., 2018; Coppola et al., 2019; Muvea
etal., 2014).
Many Trichoderma spp. are capable of actively
parasitizing the bodies of insects. The vast major-
ity of research studies carried out in this area have
reported different percentages of insect mortality. It
was reported that T. longibrachiatum and T. harzi-
anum are able to parasitize adult hemipterans such
as Bemisia tabaci Gennadius and Cimex hemipterus
L., causing mortality rates of 40% and 90% within
5 -14days, respectively (Anwar etal., 2016; Zahran
etal., 2017). Secondary metabolites (peptaibols) of
T. atroviride and T. harzianum resulted in mortality
rates of 100% in 15days in red flour beetle (Tribo-
lium castaneum Herbst), wheat aphids (Schizaphis
graminum Rondani and Diuraphis noxia Kurdj) and
the cotton aphid (Aphis gossypii Glover, Amrasca
bigutulla bigutulla Ishida) (Nawaz et al., 2020;
Rahim & Iqbal, 2019). Fungal chitinases have been
exploited to combat losses caused by insect pests.
Chitinolytic enzymes derived from T. viride were
Eur J Plant Pathol
1 3
Vol.: (0123456789)
examined in vitro and in vivo to target the peri-
trophic matrix of Lepidopteran insects (Berini
et al., 2016). These chitinases are known to attack
the insect cuticle. Field inoculation with T. harzi-
anumin the rhizosphere of maize plants resulted in
alterations in the arthropod community by activating
defense responses against herbivory. Furthermore,
research showed a reduced abundance of specific
insects under field conditions (Contreras-Cornejo
et al., 2021). Antifeedant activity of some com-
pounds, such as methyl linoleate and linoleic acid,
produced by these biocontrol fungi has been reported
against hemiptera (Kaushik et al., 2020). Recently,
a Trichoderma sp. was exploited to control a leaf-
cutting ant (Cotazo-Calambas et al., 2022). There
are some examples that advocated the activation of
resistance to insect pests by Trichoderma. This bio-
control fungus has been shown to induce jasmonic
acid (JA) mediated resistance against the scale insect
Unaspis mabilis Lit & Barbecho (Silva etal., 2019).
Similarly, activation of salicylic acid (SA) mediated
resistance in tomato plants against Bemisia tabaci
was also reported by Jafarbeigi etal. (2020). Use of
Trichoderma against various insects and its mode of
action are well documented in Table2. However, it is
necessary to note that the majority of studies exam-
ining Trichoderma as a mycopesticide were carried
out under controlled conditions. Thus, a greater num-
ber of investigations under field conditions should be
performed. Furthermore, an emphasis on the possi-
ble effects of Trichoderma on natural enemies should
be conducted simultaneously. Nevertheless, the
direct and indirect effectiveness of these fungi make
it a sustainable alternative for use in the management
of insect pests.
Bioactive compounds secreted byTrichoderma
Current research on phytoprotection has shown that
bioactive compounds play a vital role in ecofriendly
disease management systems. These compounds
consist of alkaloids, flavonoids, terpenes, phenolic
acids, salicylic acids and benzoic acid, and play a
pivotal role in host resistance induction against dif-
ferent pathogenic microbes and pests (Manganyi &
Ateba, 2020; Sharma etal., 2023). Trichoderma spp.
are known to produce a plethora of bioactive com-
pounds that includes alkenes, ketones, alcohols,
esters, monoterpenes and sesquiterpenes that induce
plant growth and reduce biotic and abiotic stress
(Bisen etal., 2016). These secondary metabolites are
involved in multifarious roles in different fields of sci-
ence and technology. Bioactive compounds auch as
Trichocaranes, Koninginins, Trichosetin, Cyclonero-
diol, Harzianolide, Trichokonin, Viridiol and 6-pen-
tyl-2H-pyran-2-one are secreted by different species of
Trichoderma and are known to regulate plant growth
(Bononi et al., 2020; Keswani et al., 2014). Several
metabolites such as Viridepyrone, Massoilactone,
Koninginins, Viridin, Viridiofungins, Harzianopyri-
done, T22 azaphilone, Harzianolide, Trichostroma-
ticins and Harzianic acid have shown antimicrobial
action and are used in biocontrol (Khan etal., 2020).
Trichoderma citrinoviride is known to produce sec-
ondary metabolites known as bisorbicillinoids that
are known to affect the feeding preference of the
aphidSchizaphis graminum Rondani (Evidente etal.,
2009). Gliotoxins, produced by Trichoderma spp.,
exhibit bioactivity against some human and plant
pathogenic fungi. Trichoderma virens produces glio-
toxin that shows an inhibitory effect against Rhizoc-
tonia bataticola, Macrophomina phaseolina, Pythium
debaryanum, Sclerotium rolfsii and Rhizoctonia solani
(Scharf etal., 2016; Shyamli etal., 2005). There are
reports that advocate the antifungal effect of Tricho-
derma metabolites against Fusarium oxysporum, Bot-
rytis fabae and other soil borne fungi under field con-
ditions (Barakat etal., 2014; Zhang etal., 2014).
A newly identified species of T. phaya-
oense increased plant height, shoot and root dry
weight values and was able to tolerate metalaxyl at
recommended dosages. In addition to its plant growth
promoting ability, this fungus effectively controlled
gummy stem blight and wilt of Cucumis melo L.
(muskmelon) (Nuangmek etal., 2021). Nevertheless,
antimicrobial compounds such as 1- pentanol, 1-hex-
anol, myristonyl pantothenate, bisabolol,
d
-Alanine,
and diethyl trisulphide, secreted by T. longibrachia-
tum, showed potential plant growth promotion and is
reported to trigger amino sugar metabolism in plants
against Macrophomina phaseolina (Sridharanet al.,
2021). It has been reported that volatile compounds
identified as alkenes, alkanes, and esters produced
by T. koningiopsis exerted biocontrol action against
fungi by suppressing plant disease and promoting
plant growth (You etal., 2022). A recent report advo-
cates that T. asperellum promotes pea growth, and
Eur J Plant Pathol
1 3
Vol:. (1234567890)
secondary metabolites play a vital role in suppression
of Globiosporangium ultimum (Moussa etal., 2023).
T. asperellumhas also been reported to promote the
growth of Allium cepa L. (onion) plants by increasing
photosynthetic activity, peroxidase activity and con-
tent of phenolic compounds (Rodríguez-Hernández
etal., 2023). In addition, Trichodenones, Trichoder-
mamide B, Harzianum A, Aurocitrin, Harziphilone,
Viridin, Virone, Viridiofungin etc. are used as anti-
cancer agents (Ebrahimi etal., 2021). Table3 depicts
the application of various bioactive compounds
produced by Trichoderma. Thus, it is evident that
the study of bioactive volatile compounds of Tricho-
derma has emerged as a frontier area of research in
recent years. Although this area of research is devel-
oping, the emergence of recent techniques has played
a vital role in the identification and characterization
of these volatile compounds. Recently, the coupling
of modern omics technology has opened up new areas
of research on these bioactive compounds.
Trichoderma bioformulations forefficient
application
The recent shift from application of synthetic fertiliz-
ers and pesticides to organic methods has brought into
focus the use of microbes that carry analogous functions
in agriculture. The bioformulations that are available in
global markets comprise of talc, liquid and secondary
metabolite based formulations. For the development of
efficient formulations, different factors such as selection
of potent antimicrobial strains, their shelf life, storage,
quality control, application, biosafety and registration are
taken into account. It has been well documented in the
above sections that several species of Trichoderma pos-
sess such biocontrol ability. For commercialization, the
mass multiplication of Trichoderma is of utmost impor-
tance. Researchers around the globe have adopted dif-
ferent formulation strategies such as the use of carboxyl
cellulose vermiculite, powders (such as talc, kaolin,
chitosan, vermiculite, ethyl cellulose, biochar, peat, and
arabic gum), alginate capsules, microencapsulation, dis-
persible granules, and seed coatings for effective delivery
of this bioagent (Lewis & Lumsden, 2001; Sriram etal.,
2011; Swaminathan etal., 2016). To address the draw-
backs of low viability and short shelf life of the biocon-
trol agent during storage, microencapsulation had been
developed. This process involves coating of the active
substances by extremely small capsules, providing the
living cells with a physical barrier against the external
environment. It has been documented that microencap-
sulation of T. harzianum in maltodextringum arabic
resulted in the highest conidial survival after spray-dry-
ing compared to nonencapsulated conidia (Muñoz-Cel-
aya et al., 2012). In another study, the development
of Trichoderma sp. formulations in encapsulated gran-
ules (CG) ensured better stability of formulations due to
good interaction between the alginate matrix and the pol-
ymers used in the formulation. It was also advocated that
this method enhanced the viable cell concentrations of
Trichoderma above 106CFU/g after 14months of stor-
age at 28°C (Locatelli etal., 2018). Further, granular and
liquid formulations of T. asperellum improved its antago-
nism against Rhizoctonia solani on corn seedlings (Her-
rera et al., 2020). Encapsulation of T. harzianum with
nanocellulose and carboxymethyl cellulose is docu-
mented to favor the fungal growth due to the extra carbon
source. These compounds are actively used as promising
materials for protecting and delivering microbial inocu-
lants (Brondi etal., 2022). Recently, Santos-Díaz et al.
(2022) described a step-by-step protocol for the stor-
age stability and partical size determination of Tricho-
dermaspp. in hydrogel beads. Encapsulation in hydrogel
beads provides an effective carrier and long-lasting bio-
product. Pradhan etal. (2022) evaluated the performance
of two types of formulations (tablet and powder based)
of Trichoderma viride against Fusarium wilt disease in
chickpea (Cicer arietinumL.). Under field conditions the
biocontrol potential of the powder based formulation was
superior for reducing wilt incidence. Table3 depicts the
recent research that advocates the use of different for-
mulations of Trichoderma and its bioefficacy. For large
scale promotion of Trichoderma formulations, it is neces-
sary to strengthen research-industry partnerships to scale
up production and delivery systems of these biocontrol
agents.
Conclusion andfuture perspectives
Trichoderma is lucratively used as potent biocontrol
agents worldwide due to its prominent antimicrobial
activity against a broad range of phytopathogens and
insects. Undoubtedly, the chemically diverse second-
ary metabolites produced by these fungi are the main
source of their efficient biocontrol potential. Mul-
tiple strategies have been adapted for development
Eur J Plant Pathol
1 3
Vol.: (0123456789)
Table 3 Application of bioactive compounds and formulations produced by Trichoderma species
Bioactive compound Trichoderma spp Effect Reference
6-pentyl-α-pyrone Trichoderma harzianum Plant metabolism and fruit quality Carillo etal., 2020
Sesquiterpenes and cyclodepsipeptides Trichoderma longibrachiatum Active against Colletotrichum lagrnarium, Bot-
rytis cinerea, Fusarium oxysporumf. sp.cuc-
umerinum and Meloidogyne incognita
Du etal., 2020
Acetonic extracts—palmitic acid and acetic acid Trichoderma virideandTrichoderma harzianum Active against Fusarium proliferatumandFusar-
ium verticillioides Yassin etal., 2021
Citric acid Trichoderma asperellum Active against Fusariumoxysporumf. sp.lyco-
persici and tomato growth promotion
Al-Askar etal., 2021
Sesquiterpenes-cedrene Trichoderma guizhouense Modulates root development Li etal., 2022
6-pentyl-α-pyrone and cyclooctanol TrichodermavirideandTrichoderma harzianum Active against Alternaria alternata Yassin etal., 2022
6-pentyl-α-pyrone Trichoderma asperellum Active against Magnaporthiopsis maydis Degani & Gordani, 2022
6-pentyl-α-pyrone, viridiofungin A, the chemical
structures of harzianic acid, iso-harzianic acid
and harzianolide
Trichodermaspp. Active against Phakopsora pachyrhizi El-Hasan etal., 2022
Phosphatases, siderophores, hydrogen cyanide,
ammonia, gibberellic acid and indole-3-acetic
acid
Trichoderma harzianum Plant growth-promoting properties Abdenaceur etal., 2022
Current studies on application of different Trichoderma formulations
Trichoderma spp Formulation technique Application Reference
Trichodermaspp. Trichogenic lipid nanoemulsion Signalling of resistance against Sclerospora
graminicola Nandini etal., 2019
Trichoderma reesei Lignin based encapsulation Efficient enzyme-responsive drug delivery sys-
tem for fungal spores
Peil etal., 2020
Trichoderma harzianum Calcium Alginate Microparticles Improved chitinolytic and cellulosic activity,
greater control against Sclerotinia sclerotiorum Maruyama etal., 2020
Trichoderma harzianum Nanocellulose/carboxymethyl cellulose nano-
composite
Better protecting and delivery of microbial
inoculants
Brondi etal., 2022
Trichoderma sp. Hydrogel based formulation Enhanced growth and yield parameters of rice Singh etal., 2022
Trichoderma asperellum Submerged liquid fermentation Yielded high microsclerotia Locatelli etal., 2022
Trichoderma asperellum Biopriming of chilli seeds with sodium alginate Enhanced efficacy in seed biopriming and toler-
ance Fusarium solani Chin etal., 2022
Trichoderma atrobrunneum Biopolymer-based emulsions Maintenance of 70–100% conidial viability after
3–6months
Martínez etal., 2023
Trichoderma asperellum Microcapsules Improved the survivability ofT. asperel-
lumconidia, biocontrol of cucumber powdery
mildew caused bySphaerotheca fuliginea
Qi etal., 2023
Eur J Plant Pathol
1 3
Vol:. (1234567890)
of biopesticide formulations of Trichoderma, such
as whole organism, spore suspension and talc-based
formulations, which have proved successful in con-
trolling various plant pathogens. However, there are
many challenges in the efficient use of Trichoderma
as a biopesticide. Its potential to be used in curative
treatments and postharvest management systems still
remains unclear. The curative property of Trichoderma
and its mechanism of biocontrol should be studied in
detail because only partial curative effects have been
so far been observed. Current research should focus
on improving the efficacy of Trichoderma-based for-
mulations under field conditions. The interaction of
Trichoderma with soil microbiome and plant micro-
biome should also be studied in depth. More studies
optimizing the time of application of formulations are
necessary to improve its biocontrol performance. In
addition, one serious challenge for future research is
understanding the pathogen recognition mechanism
involded in indirect biocontrol. Researchers should
focus on how Trichoderma assists plants in recogniz-
ing specific pathogens and effectors involved in the
activation of resistance in plants during Trichoderma-
pathogen interactions. The research on immunization
treatments of Trichoderma biomolecules should also
be strenghthened. The biosynthesis and secretion of
secondary metabolites involves the expression of dif-
ferent genes in Trichoderma, and the potential for
overexpression of these genes needs to be investigated
for further application of these compounds in vivo.
The variables that affect biocontrol performanace and
establishment of Trichoderma under field conditions,
greenhouse and in postharvest disease management
should be identified. In addition, the research on smart
methods of delivery systems should be strengthened.
Furthermore, to produce a consistent biocontrol effect
the application of these fungi should be amalgamated
with sustainable crop management practices. Conse-
quently, the development of the -omic era has opened
up new areas of research that can provide a conveni-
ent source of candidate genes for further utilization in
transgenics and crop improvement.
Acknowledgements The authors are grateful to the Depart-
ment of Plant Pathology, Dr Y S Parmar University of Horti-
culture and Forestry Solan, for continuous support.
Author’s contributions All authors have contributed equally
in manuscript preparation. All authors have read, revised, and
approved the manuscript.
Data availability Data sharing not applicable to this article
as no datasets were generated or analysed during the current
study.
Declarations
Conflict of interests The authors declare that they have no
competing interests.
References
Ab Rahman, S. S., Singh, E., Pieterse, C. M., & Schenk, P.
M. (2018). Emerging microbial biocontrol strategies for
plant pathogens. Plant Science, 267, 102–111.
Abbasi, K., Zafari, D., Wick, R., & Hamedan, I. (2017). Evalu-
ation of chitinase enzyme in fungal isolates obtained
from golden potato cyst nematode (Globoderaros-
tochiensis). Žemdirbystė (agriculture), 104(2), 179–184.
Abd-El-Khair, H., Abdel-Gaied, T. G., Mikhail, M. S., Abdel-
Alim, A. I., & El-Nasr, H. I. S. (2021). Biological control
of Pectobacterium carotovorum subsp. carotovorum, the
causal agent of bacterial soft rot in vegetables, in vitro
and in vivo tests. Bulletin of the National Research Cen-
tre, 45(1), 1–9.
Abdelkhalek, A., Al-Askar, A. A., Arishi, A. A., & Behiry, S.
I. (2022). Trichoderma hamatum strain Th23 promotes
tomato growth and induces systemic resistance against
Tobacco Mosaic Virus. Journal of Fungi, 8(3), 228.
Abdel-Shafi, S., Abdel-Gawd, S., & Sleem, E. (2013). Induc-
tion of systemic resistance and enhanced enzyme activity
by Trichoderma sp. Shmosa Tri (FJ 937359) in Squash
against Zucchini Yellow Mosaic Virus (ZYMV). Egyp-
tian Journal of Botany, 3rd International Con 17-18
April (pp. 539–558) Helwan University.
Abdenaceur, R., Farida, B. T., Mourad, D., Rima, H., Zahia,
O., & Fatma, S. H. (2022). Effective biofertilizer
Trichoderma spp. isolates with enzymatic activity and
metabolites enhancing plant growth. International
Microbiology, 25(4), 817–829.
Abelyousr, K. A. M., & Almasaudi, N. (2022). Application
of Trichoderma harzianum Strain KABOFT4 for man-
agement of tomato bacterial wilt under greenhouse
conditions. Gesunde Pflanzen, 74(2), 413–421.
Agrios, G. N. (2005). Plant Pathology. Elsevier Academic
Press.
AL-Abedy, A. N., Al-Shujairi, K. A., Al-Salami, I., Ashfaq,
M., & AL-Musawi, B. H. (2021). Genetic variation
among some isolates of tomato yellow leaf curl virus
and its control using some biological control fungi and
nanoparticles. International Journal of Agricultural
and Statistical Sciences, 17(1), 229–236.
Al-Askar, A. A., Saber, W. I., Ghoneem, K. M., Hafez, E. E.,
& Ibrahim, A. A. (2021). Crude citric acid of Tricho-
derma asperellum: tomato growth promotor and sup-
pressor of Fusarium oxysporum f. sp. lycopersici.
Plants, 10(2), 222.
Almeida, N. O., de Oliveira, C. M., Ulhoa, C. J., Cortes, M.
V. D. C. B., Júnior, M. L., & da Rocha, M. R. (2022).
Trichoderma harzianum and Trichoderma asperellum
Eur J Plant Pathol
1 3
Vol.: (0123456789)
are potential biocontrol agents of Meloidogyne javan-
ica in banana cv. Grande Naine. Biological Control,
175, 105054.
Anwar, J., & Iqbal, Z. (2017). Effect of growth conditions
on antibacterial activity of Trichoderma harzianum
against selected pathogenic bacteria. Sarhad Journal of
Agriculture, 33(4), 501–510.
Anwar, W., Subhani, M. N., Haider, M. S., Shahid, A. A.,
Mushatq, H., Rehman, M. Z., & Javed, S. (2016). First
record of Trichoderma longibrachiatum as entomopath-
ogenic fungi against Bemisia tabaci in Pakistan. Paki-
stan Journal of Phytopathology, 28(2), 287–294.
Atanasova, L., Druzhinina, I. S., Jaklitsch, W. M., Mukher-
jee, P., Horwitz, B., & Singh, U. (2013). Two hundred
Trichoderma species recognized on the basis of molec-
ular phylogeny. In P. Mukherjee, B. A. Horwitz, U. S.
Singh, M. Mukherjee, & M. Schmoll (Eds.), Tricho-
derma: biology and applications (pp. 10–42). CABI.
Babu, B. V., Kamra, A., Paul, S., & Devi, T. P. (2019). Anti-
biosis and egg parasitization in root-knot nematode.,
Meloidogyne incognita by indigenous isolates of
Trichoderma harzianum rifai, 1969 in relation to chi-
tinase and protease levels. Indian Journal of Nematol-
ogy, 49(2), 187–192.
Baiyee, B., Ito, S. I., & Sunpapao, A. (2019). Trichoderma
asperellum T1 mediated antifungal activity and induced
defense response against leaf spot fungi in lettuce (Lac-
tuca sativa L.). Physiological and Molecular Plant
Pathology, 106, 96–101.
Barakat, F. M., Abada, K. A., Abou-Zeid, N. M., & El-Gam-
mal, Y. H. E. (2014). Effect of volatile and non-volatile
compounds of Trichoderma spp. on Botrytis fabae the
causative agent of faba bean chocolate spot. American
Journal of Life Sciences, 2, 11–18.
Battaglia, D., Bossi, S., Cascone, P., Digilio, M. C., Prieto,
J. D., Fanti, P., ... & Trotta, V. (2013). Tomato below
ground–above ground interactions: Trichoderma lon-
gibrachiatum affects the performance of Macrosiphum
euphorbiae and its natural antagonists.Molecular plant-
microbe interactions,26(10), 1249-1256.
Berbert, P. S., Vieira, P. M., Cabral, G. B., Martins, E. C.,
Wulff, N. A., Ulhoa, C. J., & Aragão, F. J. (2022).
Expression of a sphingomyelinase-coding gene from
Trichoderma harzianum conferred bacterial tolerance in
tobacco. Plant Pathology, 71(5), 1048–1055.
Berini, F., Caccia, S., Franzetti, E., Congiu, T., Marinelli,
F., Casartelli, M., & Tettamanti, G. (2016). Effects of
Trichoderma viride chitinases on the peritrophic matrix
of Lepidoptera. Pest Management Science, 72(5),
980–989.
Bernard, G. C., Egnin, M., & Bonsi, C. (2017). The impact of
plant-parasitic nematodes on agriculture and methods of
control. Nematology Concepts, Diagnosis and Control,
10, 121–151.
Bisen, K., Keswani, C., Patel, J. S., Sarma, B. K., & Singh, H.
B. (2016). Trichoderma spp efficient inducers of systemic
resistance in plants. In D. K. Chaudhary & A. Verma
(Eds.), Microbial-mediated induced systemic resistance
in plants (pp. 185–195). Singapore: Springer.
Bissett, J., Szakacs, G., Nolan, C. A., Druzhinina, I., Gradinger,
C., & Kubicek, C. P. (2003). New species of Trichoderma
from Asia. Canadian Journal of Botany, 81(6), 570–586.
Bissett, J., Gams, W., Jaklitsch, W., & Samuels, G. J. (2015).
Trichoderma names in the year 2015. IMA Fungus, 6,
263–295.
Bononi, L., Chiaramonte, J. B., Pansa, C. C., Moitinho, M. A.,
& Melo, I. S. (2020). Phosphorus-solubilizing Tricho-
derma spp. from Amazon soils improves soybean plant
growth. Scientific Reports, 10(1), 2858.
Braithwaite, M., Clouston, A., Minchin, R., Yardley, J., Nieto-
Jacobo, M. F., Mendoza-Mendoza, A., Steyaert, J., Hill,
R., Marshall, J., & Stewart, A. (2016). The density-
dependent effect of initial nematode population levels on
the efficacy of Trichoderma as a bio-nematicide against
Meloidogyne hapla on tomato. Australasian Plant
Pathology, 45, 473–479.
Brondi, M., Florencio, C., Mattoso, L., Ribeiro, C., & Farinas,
C. (2022). Encapsulation of Trichoderma harzianum
with nanocellulose/carboxymethyl cellulose nanocom-
posite. Carbohydrate Polymers, 295, 119876.
Cai, F., Dou, K., Wang, P., Chenthamara, K., Chen, J., & Dru-
zhinina, I. S. (2022). The current state of Trichoderma
taxonomy and species identification. In N. Amaresan, A.
Sankaranarayanan, M. K. Dwivedi, & I. S. Druzhinina
(Eds.), Advances in Trichoderma Biology for Agricul-
tural Applications. Fungal Biology (pp. 3–35). Cham:
Springer.
Caracciolo, R., Sella, L., De Zotti, M., Bolzonello, A., Armel-
lin, M., Trainotti, L., ... & Tundo, S. (2023). Efficacy of
Trichoderma longibrachiatum Trichogin GA IV peptai-
bol analogs against the black rot pathogen Xanthomonas
campestris pv. campestris and other phytopathogenic
bacteria.Microorganisms,11(2), 480.
Carillo, P., Woo, S. L., Comite, E., El-Nakhel, C., Rouphael,
Y., Fusco, G. M., ... & Vinale, F. (2020). Application of
Trichoderma harzianum, 6-pentyl-α-pyrone and plant
biopolymer formulations modulate plant metabolism and
fruit quality of plum tomatoes.Plants,9(6), 771.
Chen, J., Li, Q. X., & Song, B. (2020). Chemical nematicides:
Recent research progress and outlook. Journal of Agri-
cultural and Food Chemistry, 68(44), 12175–12188.
Chin, J. M., Lim, Y. Y., & Ting, A. S. Y. (2022). Biopriming
chilli seeds with Trichoderma asperellum: A study on
biopolymer compatibility with seed and biocontrol agent
for disease suppression. Biological Control, 165, 104819.
Contreras-Cornejo, H. A., Macías-Rodríguez, L., del-Val, E.,
& Larsen, J. (2018). The root endophytic fungus Tricho-
derma atroviride induces foliar herbivory resistance in
maize plants. Applied Soil Ecology, 124, 45–53.
Contreras-Cornejo, H. A., Viveros-Bremauntz, F., del-Val, E.,
Macías-Rodríguez, L., López-Carmona, D. A., Alarcón,
A., ... & Larsen, J. (2021). Alterations of foliar arthropod
communities in a maize agroecosystem induced by the
root-associated fungus Trichoderma harzianum.Journal
of Pest Science,94(2), 363-374
Coppola, M., Diretto, G., Digilio, M. C., Woo, S. L., Giuliano,
G., Molisso, D., ... & Rao, R. (2019). Transcriptome
and metabolome reprogramming in tomato plants by
Trichoderma harzianum strain T22 primes and enhances
Eur J Plant Pathol
1 3
Vol:. (1234567890)
defense responses against aphids.Frontiers in Physiol-
ogy,10, 745.
Cotazo-Calambas, K. M., Niño-Castro, A., Valencia-Giraldo,
S. M., Gómez-Díaz, J. S., & Montoya-Lerma, J. (2022).
Behavioral Response of the Leaf-Cutting Ant Atta ceph-
alotes (Hymenoptera: Formicidae) to Trichoderma sp.
Journal of Insect Behavior, 35(4), 92–102.
De la Cruz-Quiroz, R., Roussos, S., Rodríguez-Herrera, R.,
Hernandez-Castillo, D., & Aguilar, C. N. (2018). Growth
inhibition of Colletotrichum gloeosporioides and Phy-
tophthora capsici by native Mexican Trichoderma
strains. Karbala International Journal of Modern Sci-
ence, 4(2), 237–243.
de Oliveira, C. M., Almeida, N. O., Côrtes, M. V. D. B., Júnior,
M. L., da Rocha, M. R., & Ulhoa, C. J. (2021). Biologi-
cal control of Pratylenchus brachyurus with isolates of
Trichoderma spp. on soybean. Biological Control, 152,
104425.
Decraemer, W., & Hunt, D. J. (2006). Structure and clas-
sification. In R. N. Perry & M. Moens (Eds.), Plant
Nematology (pp. 3–32). Wallingford, Oxfordshire: CAB
International.
Degani, O., & Gordani, A. (2022). New Antifungal Com-
pound., 6-Pentyl-α-Pyrone., against the Maize Late Wilt
Pathogen. Magnaporthiopsis maydis. Agronomy, 12(10),
2339.
del Carmen, H., Rodríguez, M., Evans, H. C., de Abreu, L.
M., de Macedo, D. M., Ndacnou, M. K., Bekele, K. B.,
& Barreto, R. W. (2021). New species and records of
Trichoderma isolated as mycoparasites and endophytes
from cultivated and wild coffee in Africa. Scientific
Reports, 11(1), 1–30.
Doehlemann, G., Ökmen, B., Zhu, W., & Sharon, A. (2017).
Plant pathogenic fungi. Microbiology Spectrum, 5(1),
5-1.
Dou, K., Lu, Z., Wu, Q., Ni, M., Yu, C., Wang, M., & Zhang,
C. (2020). MIST: A multilocus identification system for
Trichoderma. Applied and Environmental Microbiology,
86(18), e01532-e1620.
Druzhinina, I. S., Kopchinskiy, A. G., Komoń, M., Bissett,
J., Szakacs, G., & Kubicek, C. P. (2005). An oligonu-
cleotide barcode for species identification in Tricho-
derma and Hypocrea. Fungal Genetics and Biology, 42,
813–828.
Druzhinina, I. S., Kopchinskiy, A. G., & Kubicek, C. P. (2006).
The first 100 Trichoderma species characterized by
molecular data. Mycoscience, 47(2), 55–64.
Du, F. Y., Ju, G. L., Xiao, L., Zhou, Y. M., & Wu, X. (2020).
Sesquiterpenes and cyclodepsipeptides from marine-
derived fungus Trichoderma longibrachiatum and their
antagonistic activities against soil-borne pathogens.
Marine Drugs, 18(3), 165.
Dubey, V. K., & Singh, V. P. (2010). Molecular characteri-
zation of cucumber mosaic virus infecting gladiolus,
revealing its phylogeny distinct from the Indian isolate
and alike the Fny strain of CMV. Virus Genes, 41(1),
126–134.
Ebrahimi, N., Amirmahani, F., Sadeghi, B., & Ghanaatian, M.
(2021). Trichoderma longibrachiatum derived metabolite
as a potential source of anti-breast-cancer agent. Bio-
logia, 76, 1595–1601.
Elgorban, A. M., Abdel-Wahab, M. A., Bahkali, A. H., & Al-
Sum, B. A. (2014). Biocontrol of Meloidogyne javanica
on tomato plants by Hypocrea lixii (the Teleomorph of
Trichoderma harzianum). Clean-Soil Air Water, 42,
1464–1469.
Elad, Y., Chet, I., Boyle, P., & Henis, Y. (1983). Parasitism of
Trichoderma spp. on Rhizoctoniasolani and Sclerotium-
rolfsii-scanning electron microscopy and fluorescence
microscopy. Phytopathology, 73(1), 85–88.
El-Hasan, A., Walker, F., Klaiber, I., Schöne, J., Pfannstiel,
J., & Voegele, R. T. (2022). New approaches to man-
age Asian soybean rust (Phakopsora pachyrhizi) using
Trichoderma spp. or their antifungal secondary metabo-
lites. Metabolites, 12(6), 507.
Elsharkawy, M. M., Shimizu, M., Takahashi, H., Ozaki, K., &
Hyakumachi, M. (2013). Induction of systemic resistance
against cucumber mosaic virus in Arabidopsis thaliana
by Trichoderma asperellum SKT-1. The Plant Pathology
Journal, 29(2), 193.
Esmail, S. M., Omar, G. E., & Mourad, A. M. (2023). In-depth
understanding of the genetic control of stripe rust resist-
ance (Puccinia striiformis f. sp. tritici) induced in wheat
(Triticum aestivum L.) by Trichoderma asperellum T34.
Plant Disease, 107(2), 457–472.
Etim, D. O., & Onah, D. O. (2022). Trichoderma harzianum
as biocontrol agent and molecular characterisation of
papaya ringspot virus (PRSV) on Cucumeropsis mannii
in Calabar., Cross River State., Nigeria. Asian Journal of
Research in Botany, 7(4), 26–34.
Evidente, A., Andolfi, A., Cimmino, A., Ganassi, S., Altomare,
C., Favilla M., ...& Agnese Sabatini, M. (2009). Bisor-
bicillinoids produced by the fungus Trichoderma citrino-
viride affect feeding preference of the aphid Schizaphis
graminum.Journal of Chemical Ecology, 35,533-541
Fan, H., Yao, M., Wang, H., Zhao, D., Zhu, X., Wang, Y., Liu,
X., Duan, Y., & Chen, L. (2020). Isolation and effect of
Trichoderma citrinoviride Snef 1910 for the biological
control of root-knot nematode Meloidogyne incognita.
BMC Microbiology, 20, 1–11.
Ferreira, F. V., Herrmann-Andrade, A. M., Calabrese, C. D.,
Bello, F., Vázquez, D., & Musumeci, M. A. (2020).
Effectiveness of Trichoderma strains isolated from the
rhizosphere of citrus tree to control Alternaria alter-
nata., Colletotrichum gloeosporioides and Penicillium
digitatum A21 resistant to pyrimethanil in post-harvest
oranges (Citrus sinensis L.(Osbeck)). Journal of applied
microbiology, 129(3), 712–727.
Fontenelle, A. D. B., Guzzo, S. D., Lucon, C. M. M., &
Harakava, R. (2011). Growth promotion and induction of
resistance in tomato plant against Xanthomonas euvesi-
catoria and Alternariasolani by Trichoderma spp. Crop
Protection, 30(11), 1492–1500.
Gajera, H. P., Hirpara, D. G., Savaliya, D. D., & Golakiya, B.
A. (2020). Extracellular metabolomics of Trichoderma
biocontroller for antifungal action to restrain Rhizocto-
nia solani Kuhn. in cotton. Physiological and Molecular
Plant Pathology, 112, 101547.
Ganassi, S., Domenico, C. D., Altomare, C., Samuels, G. J.,
Grazioso, P., Cillo P., ... & De Cristofaro, A. (2022).
Potential of fungi of the genus Trichoderma for biocon-
trol of Philaenus spumarius, the insect vector for the
Eur J Plant Pathol
1 3
Vol.: (0123456789)
quarantine bacterium Xylella fastidosa.Pest Management
Science, 79(2), 719–728.
Ge, Y. H., Liu, K. X., Zhang, J. X., Mu, S. Z., & Hao, X. J.
(2012). The limonoids and their antitobacco mosaic
virus (TMV) activities from Munronia unifoliolata Oliv.
Journal of Agricultural and Food Chemistry, 60(17),
4289–4295.
Ghorbanpour, A., Salimi, A., Ghanbary, M. A. T., Pirdashti,
H., & Dehestani, A. (2018). The effect of Trichoderma
harzianum in mitigating low temperature stress in tomato
(Solanum lycopersicum L.) plants. Scientia Horticultu-
rae, 230, 134–141.
Ghosh, S. K., & Pal, S. (2016). Entomopathogenic potential of
Trichoderma longibrachiatum and its comparative evalu-
ation with malathion against the insect pest Leucinodes
orbonalis. Environmental Monitoring and Assessment,
188(1), 1–7.
Górzyńska, K., Ślachetka, M., Ryszka, P., Turnau, K., Płachno,
B. J., & Lembicz, M. (2018). Incidence, identification,
and mycoparasitic activity of Clonostachys epichloë, a
hyperparasite of the fungal endophyte Epichloë typhina.
Plant Disease, 102(10), 1973–1980.
Goverse, A., & Smant, G. (2014). The activation and suppres-
sion of plant innate immunity by parasitic nematodes.
Annual Review of Phytopathology, 52, 243–265.
Guigón López, C., Muñoz Castellanos, L. N., Flores Ortiz,
N. A., & González González, J. A. (2019). Control of
powdery mildew (Leveillula taurica) using Trichoderma
asperellum and Metarhizium anisopliae in different pep-
per types. BioControl, 64(1), 77–89.
Guler, N. S., Pehlivan, N., Karaoglu, S. A., Guzel, S., &
Bozdeveci, A. (2016). Trichoderma atroviride ID20G
inoculation ameliorates drought stress-induced damages
by improving antioxidant defence in maize seedlings.
Acta Physiologiae Plantarum, 38, 132.
Guo, R., Ji, S., Wang, Z., Zhang, H., Wang, Y., & Liu, Z.
(2021a). Trichoderma asperellum xylanases promote
growth and induce resistance in poplar. Microbiological
Research, 248, 12676.
Guo, Y., Fan, Z., Yi, X., Zhang, Y., Khan, R. A. A., & Zhou,
Z. (2021b). Sustainable management of soil-borne bac-
terium Ralstonia solanacearum in vitro and in vivo
through fungal metabolites of different Trichoderma
spp. Sustainability, 13(3), 1491.
Hajji-Hedfi, L., Khaled, E. B., Arcos, S. C., Robertson, L,
Ciordia, S, Gonzalez, M. R, ... & Navas A. (2021).
Insights into nematode biocontrol potential through
biological and proteomics analysis of the fungus
Trichoderma viride. In Euro-Mediterranean Confer-
ence for Environmental Integration (pp.1327–1333)
Springer Cham.
Halifu, S., Deng, X., Song, X., & Song, R. (2019). Effects of
two Trichoderma strains on plant growth, rhizosphere
soil nutrients and fungal community of Pinus sylvestris
var. mongolica annual seedlings. Forests, 10, 758.
Halifu, S., Deng, X., Song, X., Song, R., & Liang, X. (2020).
Inhibitory mechanism of Trichoderma virens ZT05 on
Rhizoctonia solani. Plants, 9(7), 912.
Harman, G. E., Howell, C. R., Viterbo, A., Chet, I., & Lorito,
M. (2004). Trichoderma species—opportunistic,
avirulent plant symbionts. Nature Reviews Microbiology,
2(1), 43–56.
Hebert, P. D. N., Cywinska, A., Ball, S. L., & de Waard, J.
R. (2003). Biological identifications through DNA bar-
codes. Proceedings of the Royal Society of London.
Series B: Biological Sciences, 270(1512), 313–321.
Heflish, A. A., Abdelkhalek, A., Al-Askar, A. A., & Behiry, S.
I. (2021). Protective and curative effects of Trichoderma
asperelloides Ta41 on tomato root rot caused by Rhizoc-
tonia solani Rs33. Agronomy, 11(6), 1162.
Hermosa, R., Viterbo, A., Chet, I., & Monte, E. (2012). Plant-
beneficial effects of Trichoderma and of its genes. Micro-
biology, 158, 17–25.
Herrera, W., Valbuena, O., & Pavone-Maniscalco, D. (2020).
Formulation of Trichoderma asperellum TV190 for bio-
logical control of Rhizoctonia solani on corn seedlings.
Egyptian Journal of Biological Pest Control, 30, 1–8.
Hewedy, O. A., Abdel, Lateif K. S., Seleiman, M. F., Shami,
A., Albarakaty, F. M., & M El-Meihy, R. (2020). Phylo-
genetic diversity of Trichoderma strains and their antago-
nistic potential against soil-borne pathogens under stress
conditions. Biology, 9(8), 189.
Vijayakumar, N., & Alagar, S. (2017). Consequence of chi-
tinase from Trichoderma viride integrated feed on diges-
tive enzymes in Corcyra cephalonica (Stainton) and anti-
microbial potential. Biosciences Biotechnology Research
Asia, 14(2), 513–519.
Inayati, A., Sulistyowati, L., Aini, L. Q., & Yusnawan, E.
(2020). Mycoparasitic activity of indigenous Tricho-
derma virens strains against mungbean soil borne patho-
gen Rhizoctonia solani: Hyperparasite and hydrolytic
enzyme production. AGRIVITA. Journal of Agricultural
Science, 42(2), 229–242.
Index Fungorum. Retrieved November 20, 2022 from http://
www. index fungo rum. org/ Names/ Names. asp
Islam, M. R., Chowdhury, R., Roy, A. S., Islam, M. N., Mita,
M. M., Bashar, S., ... & Latif, M. A. (2023). Native
Trichoderma induced the defense-related enzymes and
genes in rice against Xanthomonas oryzae pv. oryzae
(Xoo).Plants,12(9), 1864.
Jafarbeigi, F., Samih, M. A., Alaei, H., & Shirani, H. (2020).
Induced tomato resistance against Bemisia tabaci trig-
gered by salicylic acid., β-aminobutyric acid., and
Trichoderma. Neotropical Entomology, 49, 456–467.
Jaklitsch, W. M. (2009). European Species of Hypocrea Part
I. The Green-Spored Species. Studies in Mycology, 63,
1–91.
Jatav, P., Ahirwar, S. S., Gupta, A., Kushwaha, K., & Jatav, S.
(2018). Antagonistic activity of cellulase enzyme pro-
duced by Trichoderma viride against Xanthomonas citri.
Indian Journal of Agricultural Research, 52(5), 497–504.
Jones, J. T., Haegeman, A., Danchin, E. G., Gaur, H. S.,
Helder, J., Jones, M. G., ... & Perry, R. N. (2013). Top
10 plant‐parasitic nematodes in molecular plant pathol-
ogy.Molecular Plant Pathology, 14(9), 946–961.
Kamala, T., Devi, S. I., Sharma, K. C., & Kennedy, K. (2015).
Phylogeny and taxonomical investigation of Trichoderma
spp. from Indian region of Indo-Burma biodiversity hot
spot region with special reference to Manipur. BioMed
Research International, 2015, 1–21.
Eur J Plant Pathol
1 3
Vol:. (1234567890)
Kaushik, N., Díaz, C. E., Chhipa, H., Julio, L. F., Andrés, M.
F., & González-Coloma, A. (2020). Chemical composi-
tion of an aphid antifeedant extract from an endophytic
fungus, Trichoderma sp. EFI671. Microorganisms, 8(3),
420.
Keswani, C., Mishra, S., Sarma, B. K., Singh, S. P., & Singh,
H. B. (2014). Unraveling the efficient applications of sec-
ondary metabolites of various Trichoderma spp. Applied
Microbiology and Biotechnology, 98, 533–544.
Khan, R. A. A., Najeeb, S., Hussain, S., Xie, B., & Li, Y.
(2020). Bioactive secondary metabolites from Tricho-
derma spp. against phytopathogenic fungi. Microorgan-
isms, 8(6), 817.
Kiarie, S., Nyasani, J. O., Gohole, L. S., Maniania, N. K., &
Subramanian, S. (2020). Impact of fungal endophyte col-
onization of maize (Zea mays L.) on induced resistance
to thrips-and aphid-transmitted viruses. Plants, 9(4), 416.
Kiriga, A. W., Haukeland, S., Kariuki, G. M., Coyne, D. L.,
& Beek, N. V. (2018). Effect of Trichoderma spp. and
Purpureocillium lilacinum on Meloidogyne javanica in
commercial pineapple production in Kenya. Biological
Control, 119, 27–32.
Klaram, R., Jantasorn, A., & Dethoup, T. (2022). Efficacy of
marine antagonist Trichoderma spp. as halo-tolerant bio-
fungicide in controlling rice diseases and yield improve-
ment. Biological Control, 172, 104985.
Konappa, N., Krishnamurthy, S., Siddaiah, C. N., Ramachan-
drappa, N. S., & Chowdappa, S. (2018). Evaluation of
biological efficacy of Trichoderma asperellum against
tomato bacterial wilt caused by Ralstonia solanacearum.
Egyptian Journal of Biological Pest Control, 28(1),
1–11.
Laib, D. E., Benzara, A., Akkal, S., & Bensouici, C. (2020).
The anti-acetylcholinesterase, insecticidal and antifungal
activities of the entophytic fungus Trichoderma sp. iso-
lated from Ricinus communis L. against Locusta migra-
toria. Acta Scientifica Naturalis, 7(1), 112–125.
Lang, B. Y., Li, J., Zhou, X. X., Chen, Y. H., Yang, Y. H., Li,
X. N., Zeng, Y., & Zhao, P. J. (2015). Koninginins L and
M, two polyketides from Trichoderma koningii 8662.
Phytochemistry Letters, 11, 1–4.
Latz, M. A., Jensen, B., Collinge, D. B., & Jørgensen, H. J.
(2018). Endophytic fungi as biocontrol agents: Elucidat-
ing mechanisms in disease suppression. Plant Ecology
and Diversity, 11(5–6), 555–567.
Lazazzara, V., Vicelli, B., Bueschl, C., Parich, A., Pertot, I.,
Schuhmacher, R., & Perazzolli, M. (2021). Trichoderma
spp. volatile organic compounds protect grapevine plants
by activating defense-related processes against downy
mildew. Physiologia plantarum, 172(4), 1950–1965.
Lewis, J. A., & Lumsden, R. D. (2001). Biocontrol of damp-
ing-off of greenhouse-grown crops caused by Rhizocto-
nia solani with a formulation of Trichoderma spp. Crop
Protection, 20(1), 49–56.
Li, G., Zhang, K., Xu, J., Dong, J., & Liu, Y. (2007). Nemati-
cidal substances from fungi. Recent Patents on Biotech-
nology, 1, 212–233.
Li, Y., Shao, J., Fu Y., Chen, Y., Wang, H., & Xu, Z, ... Zhang,
R. (2022). The volatile cedrene from Trichoderma
guizhouense modulates Arabidopsis root development
through auxin transport and signalling. Plant, Cell and
Environment, 45(3), 969–984.
Litwin, A., Nowak, M., & Różalska, S. (2020). Entomopath-
ogenic fungi: unconventional applications. Reviews
in Environmental Science and Bio/Technology, 19(1),
23–42.
Locatelli, G. O., dos Santos, G. F., Botelho, P. S., Finkler, C.
L. L., & Bueno, L. A. (2018). Development of Tricho-
derma sp. formulations in encapsulated granules (CG)
and evaluation of conidia shelf-life. Biological Control,
117, 21–29.
Locatelli, G. O., Pimentel, M. F., Bueno, L. A., Junior, M. L.,
Mascarin, G. M., & Finkler, C. L. L. (2022). Production
of microsclerotia by Trichoderma asperellum through
submerged liquid fermentation using low-cost nitrogen
and carbon sources. Biocatalysis and Agricultural Bio-
technology, 44, 102455.
López-Bucio, J., Pelagio-Flores, R., & Herrera-Estrella, A.
(2015). Trichoderma as biostimulant: Exploiting the mul-
tilevel properties of a plant beneficial fungus. Scientia
Horticulturae, 196, 109–123.
Mai, W. F., & Abawi, G. S. (1987). Interactions among root-
knot nematodes and Fusarium wilt fungi on host plants.
Annual Review Phytopathology, 25, 317–338.
Manganyi, M. C., & Ateba, C. N. (2020). Untapped potentials
of endophytic fungi: A review of novel bioactive com-
pounds with biological applications. Microorganisms,
8(12), 1934.
Manivel, B. S., & Rajkumar, S. G. (2018). Mycopesticides:
Fungal based pesticides for sustainable agriculture. In P.
Gehlot & J. Singh (Eds.), Fungi and their Role in Sus-
tainable Development: Current Perspectives (pp. 183–
203). Singapore: Springer.
Martínez, Y., Heeb, M., Kalač, T., Gholam, Z., Schwarze, F.
W., Nyström, G., & De France K. (2023). Biopolymer-
based emulsions for the stabilization of Trichoderma
atrobrunneum conidia for biological control. Applied
Microbiology and Biotechnology, 107(4), 1465–1476.
Martínez-Medina, A., Fernandez, I., Lok, G. B., Pozo, M.
J., Pieterse, C. M., & Van Wees, S. C. (2017). Shifting
from priming of salicylic acid-to jasmonic acid-regulated
defences by Trichoderma protects tomato against the root
knot nematode Meloidogyne incognita. New phytologist,
213(3), 1363–1377.
Maruyama, C. R., Bilesky-José, N., de Lima, R., & Fraceto, L.
F. (2020). Encapsulation of Trichoderma harzianum pre-
serves enzymatic activity and enhances the potential for
biological control. Frontiers in Bioengineering and Bio-
technology, 8, 225.
Mastouri, F., Bjorkman, T., & Harman, G. E. (2012). Tricho-
derma harzianum enhances antioxidant defense of
tomato seedlings and resistance to water deficit. Molecu-
lar Plant-Microbe Interactions, 25, 1264–1271.
Medeiros, H. A. D., Araújo Filho, J. V. D., Freitas, L. G. D.,
Castillo, P., Rubio, M. B., Hermosa, R., & Monte, E.
(2017). Tomato progeny inherit resistance to the nema-
tode Meloidogyne javanica linked to plant growth
induced by the biocontrol fungus Trichoderma atrovir-
ide. Scientific reports, 7(1), 1–13.
Mejía, C., Ardila, H. D., Espinel, C., Brandão, P. F., & Vil-
lamizar, L. (2021). Use of Trichoderma koningiopsis
Eur J Plant Pathol
1 3
Vol.: (0123456789)
chitinase to enhance the insecticidal activity of Beau-
veria bassiana against Diatraea saccharalis. Journal of
Basic Microbiology, 61(9), 814–824.
Mohamed, B. F., Sallam, N., Alamri, S. A., Abo-Elyousr, K.
A., Mostafa, Y. S., & Hashem, M. (2020). Approving
the biocontrol method of potato wilt caused by Ralstonia
solanacearum (Smith) using Enterobacter cloacae PS14
and Trichoderma asperellum T34. Egyptian Journal of
Biological Pest Control, 30(1), 1–13.
Moo-Koh, F. A., Cristóbal-Alejo, J., Andrés, M. F., Martín,
J., Reyes, F., Tun-Suárez, J. M., & Gamboa-Angulo, M.
(2022). In vitro assessment of organic and residual frac-
tions of nematicidal culture filtrates from thirteen tropi-
cal Trichoderma strains and metabolic profiles of most-
active. Journal of Fungi, 8(1), 82.
Moosavi, M. R., & Zare, R. (2020). Fungi as biological control
agents of plant-parasitic nematodes. In J. M. Mérillon &
K. G. Ramawat (Eds.), Plant defence: biological control
(2nd ed., pp. 333–384). Springer.
Moussa, Z., Alanazi, Y. F., Khateb, A. M., Eldadamony, N. M.,
Ismail, M. M., Saber, W. I., & Darwish, D. B. E. (2023).
Domiciliation of Trichoderma asperellum suppresses
Globiosporangium ultimum and promotes pea growth,
ultrastructure, and metabolic features. Microorganisms,
11(1), 198.
Mukherjee, M., Mukherjee, P. K., Horwitz, B. A., Zachow,
C., Berg, G., & Zeilinger, S. (2012). Trichoderma-plant-
pathogen interactions: Advances in genetics of biological
control. Indian Journal of Microbiology, 53, 522–529.
Mukherjee, P. K., Horwitz, B. A., Herrera-Estrella, A.,
Schmoll, M., & Kenerley, C. M. (2013). Trichoderma
research in the genome era. Annual Review of Phytopa-
thology, 51(1), 105–129.
Mukhopadhyay, R., & Kumar, D. (2020). Trichoderma: A ben-
eficial antifungal agent and insights into its mechanism
of biocontrol potential. Egyptian Journal of Biological
Pest Control, 30(1), 1–8.
Muñoz-Celaya, A. L., Ortiz-García, M., Vernon-Carter, E. J.,
Jauregui-Rincón, J., Galindo, E., & Serrano-Carreón, L.
(2012). Spray-drying microencapsulation of Trichoderma
harzianum conidias in carbohydrate polymers matrices.
Carbohydrate Polymers, 88(4), 1141–1148.
Muvea, A. M., Meyhöfer, R., Subramanian, S., Poehling, H.
M., Ekesi, S., & Maniania, N. K. (2014). Colonization of
onions by endophytic fungi and their impacts on the biol-
ogy of Thrips tabaci. PLoS ONE, 9(9), e108242.
Naeimi, S., Okhovvat, S. M., Javan-Nikkhah, M., Vágvölgyi,
C., Khosravi, V., & Kredics, L. (2010). Biological con-
trol of Rhizoctonia solani AG1–1A., the causal agent of
rice sheath blight with Trichoderma strains. Phytopatho-
logia Mediterranea, 49(3), 287–300.
Nandini, B., Puttaswamy, H., Prakash, H. S., Adhikari, S.,
Jogaiah, S., & Nagaraja, G. (2019). Elicitation of novel
trichogenic-lipid nanoemulsion signaling resistance
against pearl millet downy mildew disease. Biomole-
cules, 10(1), 25.
Nawaz, A., Gogi, M. D., Naveed, M., Arshad, M., Sufyan, M.,
Binyameen, M., ... & Ali, H. (2020). In vivo and in vitro
assessment of Trichoderma species and Bacillus thur-
ingiensis integration to mitigate insect pests of brinjal
(Solanum melongena L.).Egyptian Journal of Biological
Pest Control,30(1), 1-7.
Neumann, N. K., Stoppacher, N., Zeilinger, S., Degenkolb,
T., Bruckner, H., & Schuhmacher, R. (2015). The pep-
taibiotics database—A comprehensive online resource.
Chemistry & Biodiversity, 12(5), 743–751.
Nicol, J. M., Turner, S. J., Coyne, D. L., Nijs, L. D., Hockland,
S., & Maafi, Z. T. (2011). Current nematode threats to
world agriculture. In J. Jones, G. Gheysen, & C. Fenoll
(Eds.), Genomics and molecular genetics of plant-nema-
tode interactions (pp. 21–43). Springer.
Nicolopoulos-Stamati, P., Maipas, S., Kotampasi, C., Stamatis,
P., & Hens, L. (2016). Chemical pesticides and human
health: The urgent need for a new concept in agriculture.
Frontiers in Public Health, 4, 1–8.
Nuangmek, W., Aiduang, W., Kumla, J., Lumyong, S., &
Suwannarach, N. (2021). Evaluation of a newly identified
endophytic fungus, Trichoderma phayaoense for plant
growth promotion and biological control of gummy stem
blight and wilt of muskmelon. Frontiers in Microbiology,
12, 634772.
Ons, L., Bylemans, D., Thevissen, K., & Cammue, B. P.
(2020). Combining biocontrol agents with chemical fun-
gicides for integrated plant fungal disease control. Micro-
organisms, 8(12), 1930.
Padovani, L., Trevisan, M., & Capri, E. (2004). A calcula-
tion procedure to assess potential environmental risk of
pesticides at the farm level. Ecological Indicators, 4(2),
111–123.
Papaianni, M., Ricciardelli, A., Fulgione, A., d’Errico, G.,
Zoina, A., Lorito, M., ... & Capparelli, R. (2020). Antibi-
ofilm Activity of a Trichoderma Metabolite against Xan-
thomonas campestris pv. campestris., Alone and in Asso-
ciation with a Phage.Microorganisms,8(5), 620.
Peil, S., Beckers, S. J., Fischer, J., & Wurm, F. R. (2020). Bio-
degradable, lignin-based encapsulation enables deliv-
ery of Trichoderma reesei with programmed enzymatic
release against grapevine trunk diseases. Materials Today
Bio, 7, 100061.
Persoon, C. H. (1794). Disposita methodica fungorum.
Römer’s Neues Mag. Bot, 1, 81–128.
Pocurull, M., Fullana, A.M., Ferro, M., Valero, P., Escudero,
N., Saus, E., ... & Sorribas, F. J. (2020). Commercial
formulates of Trichoderma induce systemic plant resist-
ance to Meloidogyne incognita in tomato and the effect
is additive to that of the Mi-1.2 resistance gene.Frontiers
in Microbiology,10, 3042.
Pradhan, P. C., Mukhopadhyay, A., Kumar, R., Kundu, A.,
Patanjali, N., Dutta, A., ... & Singh, A. (2022). Perfor-
mance appraisal of Trichoderma viride based novel tablet
and powder formulations for management of Fusarium
wilt disease in chickpea.Frontiers in Plant Science,13,
990392.
Purwantisari, S., Priyatmojo, A., Sancayaningsih, R. P., Kasi-
amdari, R. S., & Budihardjo, K. (2018). Systemic induc-
ing resistance against late blight by applying antagonist
Trichoderma viride. Journal of Physics: Conference
Series, 1025(1), 012053.
Qi, Q., Fan, C., Wu, H., Sun, L., & Cao, C. (2023). Preparation
of Trichoderma asperellum microcapsules and biocontrol
Eur J Plant Pathol
1 3
Vol:. (1234567890)
of cucumber powdery mildew. Microbiology Spectrum,
11(3), e05084–22.
Qin, W. T., & Zhuang, W. Y. (2016). Seven wood-inhabiting
new species of the genus Trichoderma (Fungi., Ascomy-
cota) in Viride clade. Scientific Reports, 6(1), 1–15.
Rahim, S., & Iqbal, M. (2019). Exploring enhanced insecti-
cidal activity of mycelial extract of Trichoderma harzi-
anum against Diuraphis noxia and Tribolium castaneum.
Sarhad Journal of Agriculture, 35(3), 757–762.
Raja, H. A., Miller, A. N., Pearce, C. J., & Oberlies, N. H.
(2017). Fungal identification using molecular tools: A
primer for the natural products research community.
Journal of Natural Products, 80(3), 756–770.
Rajani, P., Rajasekaran, C., Vasanthakumari, M. M., Olsson, S.
B., Ravikanth, G., & Shaanker, R. U. (2021). Inhibition
of plant pathogenic fungi by endophytic Trichoderma
spp. through mycoparasitism and volatile organic com-
pounds. Microbiological Research, 242, 126595.
Raymaekers, K., Ponet, L., Holtappels, D., Berckmans, B., &
Cammue, B. P. (2020). Screening for novel biocontrol
agents applicable in plant disease management–a review.
Biological Control, 144, 104240.
Redda, E. T., Ma, J., Mei, J., Li, M., Wu, B., & Jiang, X.
(2018). Antagonistic potential of different isolates of
Trichoderma against Fusarium oxysporum, Rhizoctonia
solani and Botrytis cinerea. European Journal of Experi-
mental Biology, 8(2), 1–8.
Rochal, K.K. L., Pierre, E., Diane, Y.Y., Sahu, K. P., Vanessa,
N. D., Herman, K. W.T., ... & Louise, N. W. (2021). Bio-
logical elicitor potential of endospheric Trichoderma and
derived consortia against pepper (Capsicum annuum L.)
leaf curl virus. Archives of Phytopathology and Plant
Protection,54(19–20), 1926–1952.
Rodríguez-González, Á., Mayo, S., González-López, Ó., Rei-
noso, B., Gutierrez, S., & Casquero, P. A. (2017). Inhibi-
tory activity of Beauveria bassiana and Trichoderma spp.
on the insect pests Xylotrechus arvicola (Coleoptera:
Cerambycidae) and Acanthoscelides obtectus (Coleop-
tera: Chrisomelidae: Bruchinae). Environmental Moni-
toring and Assessment, 189(1), 1–8.
Rodríguez-Hernández, A. A., Herrera-Alvarez, M., Zapata-
Sarmiento, D. H., Becerra-Martínez, E., Rodríguez-
Monroy, M., & Sepúlveda-Jiménez, G. (2023).Tricho-
derma asperellum promotes the development and
antioxidant activity of white onion (Allium cepa L.)
plants. Horticulture, Environment, and Biotechnology,
64(1), 25–39.
Ruiz-Gómez, F. J., & Miguel-Rojas, C. (2021). Antagonistic
potential of native Trichoderma spp. against Phytoph-
thora cinnamomi in the control of holm oak decline in
Dehesas ecosystems. Forests, 12(7), 945.
Rush, T. A., Shrestha, H. K., Gopalakrishnan, M. M., Spangler,
M. K., Ellis, J. C., Labbé, J. L., & Abraham, P. E. (2021).
Bioprospecting Trichoderma: A systematic roadmap
to screen genomes and natural products for biocontrol
applications. Frontiers in Fungal Biology, 2, 41.
Ryu, S.M., Lee, H. M., Song, E. G., Seo, Y. H., Lee, J., Guo,
Y., ... & Lee, D. (2017). Antiviral activities of tri-
chothecenes isolated from Trichoderma albolutescens
against pepper mottle virus.Journal of agricultural and
food chemistry,65(21), 4273–4279.
Safari Motlagh, M. R., Jahangiri, B., Kulus, D., Tymoszuk, A.,
& Kaviani, B. (2022). Endophytic fungi as potential bio-
control agents against Rhizoctonia solani JG Kühn, the
causal agent of rice sheath blight disease. Biology, 11(9),
1282.
Sahebani, N., & Hadavi, N. (2008). Biological control of the
root-knot nematode Meloidogyne javanica by Tricho-
derma harzianum. Soil Biology and Biochemistry, 40(8),
2016–2020.
Saikia, R., Paswan, R. R., Farwaha, N., & Borah, B. K. (2022).
Plant viruses: Factors involved in emergence and recent
advances in their management. In S. Roy, P. Mathur, A.
P. Chakraborty, & S. P. Saha (Eds.), Plant Stress: Chal-
lenges and Management in the New Decade (pp. 29–55).
Bangalore: Springer.
Sala, A., Barrena, R., Artola, A., & Sánchez, A. (2019). Cur-
rent developments in the production of fungal biological
control agents by solid-state fermentation using organic
solid waste. Critical Reviews in Environmental Science
and Technology, 49(8), 655–694.
Salwan, R., Sharma, A., Kaur, R., Sharma, R., & Sharma, V.
(2022). The riddles of Trichoderma induced plant immu-
nity. Biological Control, 174, 105037.
Sanchez, A. D., Ousset, M. J., & Sosa, M. C. (2019). Bio-
logical control of Phytophthora collar rot of pear using
regional Trichoderma strains with multiple mechanisms.
Biological Control, 135, 124–134.
Sanjeev, K., Manibhushan, T., & Archana, R. (2014). Tricho-
derma: Mass production, formulation, quality control,
delivery and its scope in commercialization in India for
the management of plant diseases. African Journal of
Agricultural Research, 9(53), 3838–3852.
Santos-Díaz, A., Uribe-Gutiérrez, L., & Cruz-Barrera, M.
(2022). High-throughput assessments for storage stabil-
ity, in vitro release, and particle size of encapsulated bio-
control fungi in hydrogel Beads. Current Protocols, 2(7),
e486.
Sawant, I. S., Wadkar, P. N., Ghule, S. B., Rajguru, Y. R.,
Salunkhe, V. P., & Sawant, S. D. (2017). Enhanced bio-
logical control of powdery mildew in vineyards by inte-
grating a strain of Trichoderma afroharzianum with sul-
phur. Biological Control, 114, 133–143.
Sayed, M., Abdel-rahman, T., Ragab, A., & Abdellatif, A.
(2019). Biocontrol of root-knot nematode Meloidogyne
incognita by Chitinolytic Trichoderma spp. Egyptian
Journal of Agronematology, 18(1), 30–47.
Scharf, D. H., Brakhage, A. A., & Mukherjee, P. K. (2016).
Gliotoxine bane or boon? Environmental Microbiology,
18, 1096–1109.
Scholthof, K. B. G., Adkins, S., Czosnek. H., Palukaitis, P.,
Jacquot, E., Hohn, T., ... & Foster G. D. (2011). Top 10
plant viruses in molecular plant pathology. Molecular
plant pathology, 12(9), 938–954.
Seinhorst, J. W. (1970). Dynamics of plant parasitic nema-
todes. Rev. Phytopath., 8, 131–156.
Shah, M. M., & Afiya, H. (2019). Introductory chapter: identi-
fication and isolation of Trichoderma spp.-Their signifi-
cance in agriculture, human health, industrial and envi-
ronmental application. In M. S. Mohammad, S. Umar, &
R. B. Tijjani (Eds.), Trichoderma-The Most Widely Used
Fungicide (pp. 1–12). IntechOpen.
Eur J Plant Pathol
1 3
Vol.: (0123456789)
Sharma, A., Gupta, A. K., Khosla, K., Mahajan, R., &
Mahajan, P. K. (2017). Antagonistic potential of native
agrocin-producing non-pathogenic Agrobacterium tume-
faciens strain UHFBA-218 to control crown gall in
peach. Phytoprotection, 97(1), 1–1.
Sharma, A., Kumar, V., Shahzad, B., Tanveer, M., Sidhu, G.
P. S., Handa, N., & Thukral, A. K. (2019). Worldwide
pesticide usage and its impacts on ecosystem. SN Applied
Sciences, 1(11), 1–16.
Sharma, A., Gupta, A. K., & Devi, B. (2023). Current trends
in management of bacterial pathogens infecting plants.
Antonie van Leeuwenhoek, 116(4), 303–326.
Sharon, E., Chet, I., & Spiegel, Y. (2011). Trichoderma as a
biological control agent. In K. Davies & Y. Spiegel
(Eds.), Biological Control of Plant-Parasitic Nematodes:
Building Coherence between Microbial Ecology and
Molecular Mechanisms (pp. 183–201). Springer.
Shi, X. S., Li, H. L., Li, X. M., Wang, D. J., Li, X., Meng, L.
H., Zhou, X. W., & Wang, B. G. (2020). Highly oxygen-
ated polyketides produced by Trichoderma koningiopsis
QA-3., an endophytic fungus obtained from the fresh
roots of the medicinal plant. Artemisia argyi. Bioorganic
Chemistry, 94, 103448.
Shoresh, M., Harman, G. E., & Mastoury, F. (2010). Induced
systemic resistance and plant response to fungal biocon-
trol agents. Annual Review of Phytopathology, 48, 21–43.
Shyamli, S., Prem, D., Rs, T., & Atar, S. (2005). Production
and antifungal activity of secondary metabolites of
Trichoderma virens. Pesticide Research Journal, 17,
26–29.
Siebatcheu, E. C., Wetadieu, D., Youassi, Y.O., Bedine, B. M.
A., Bedane, K. G., Tchameni., N. S., &Sameza, M. L.
(2023). Secondary metabolites from an endophytic fun-
gus Trichoderma erinaceum with antimicrobial activity
towards Pythium ultimum. Natural Product Research,
37(4), 657–662.
Silva, B. B., Banaay, C. G., & Salamanez, K. (2019). Tricho-
derma-induced systemic resistance against the scale
insect (Unaspis mabilis Lit & Barbecho) in lanzones
(Lansium domesticum Corr.). Agriculture & Forestry,
65(2), 59–78.
Sindhu, S. S., Sehrawat, A., Sharma, R., & Khandelwal, A.
(2017). Biological control of insect pests for sustainable
agriculture. In T. Adhya, B. Mishra, K. Annapurna, D.
Verma, & U. Kumar (Eds.), Advances in Soil Microbiol-
ogy Recent Trends and Future Prospects (pp. 189–218).
Singapore: Advances in Soil Microbiology: Recent
Trends and Future Prospects. Microorganisms for
Sustainability.
Singh, U. B., Singh, S., Malviya, D., Chaurasia, R., &
Imran, M. R. J. (2017). Harnessing biocontrol poten-
tial of Trichoderma harzianum for control of Meloi-
dogyne incognita in tomato. Indian Phytopathology, 70,
331–335.
Singh, B. N., Dwivedi, P., Sarma, B. K., & Singh, H. B. (2019).
Trichoderma asperellum T42 induces local defense
against Xanthomonas oryzae pv. oryzae under nitrate and
ammonium nutrients in tobacco. RSC advances, 9(68),
39793–39810.
Singh, R. K., Jatav, H. S., Lakpale, R., Khan, M., Rajput, V.
D., & Minkina, T. (2022). Hydrogel-based Trichoderma
formulation effects on different varieties of rice under
rainfed condition of Indo-Gangetic Plains. Environment
Development and Sustainability, 24(5), 7035–7056.
Siphathele, S., Lucy, N. M., Divine, Y. S., & Teresa, A. C.
(2018). Quorum sensing in gram-negative plant patho-
genic bacteria. In N. K. Josphert (Ed.), Advances in Plant
Pathology (pp. 67–89). Intech Open. https:// doi. org/ 10.
5772/ intec hopen. 78003
Smirnova, I. P., Karimova, E. V., & Shneider, Y. A. (2017).
Antibacterial activity of L-lysine-α-oxidase from the
Trichoderma. Bulletin of Experimental Biology and Med-
icine, 163(6), 777–779.
Solanki, M. K., Singh, N., Singh, R. K., Singh, P., Srivas-
tava, A. K., etal. (2011). Plant defense activation and
management of tomato root rot by a chitin-fortified
Trichoderma/Hypocrea formulation. Phytoparasitica, 39,
471–481.
Sood, M., Kapoor, D., Kumar, V., Sheteiwy, M. S., Ram-
akrishnan, M., Landi, M., Araniti, F., & Sharma, A.
(2020). Trichoderma: The “secrets” of a multitalented
biocontrol agent. Plants, 9, 762.
Sreenivasaprasad, S., & Manibhushanrao, K. (1990). Antago-
nistic potential of Gliocladium virens and Trichoderma
longibrachiatum to phytopathogenic fungi. Mycopatho-
logia, 109(1), 19–26.
Sridharan, A. P., Sugitha, T., Karthikeyan, G., Nakkeeran, S.,
& Sivakumar, U. (2021). Metabolites of Trichoderma
longibrachiatum EF5 inhibits soil borne pathogen Mac-
rophomina phaseolina by triggering amino sugar metab-
olism. Microbial Pathogenesis, 150, 104714.
Sriram, S., Roopa, K. P., & Savitha, M. J. (2011). Extended
shelf-life of liquid fermentation derived talc formulations
of Trichoderma harzianum with the addition of glyc-
erol in the production medium. Crop Protection, 30(10),
1334–1339.
Srivastava, C., & Subramanian, S. (2016). Storage insect pests
and their damage symptoms: An overview. Indian Jour-
nal of Entomology, 78, 53–58.
Sulaiman, M. M., Yass, S. T. A., Aish, A. A., Basheer, L.,
Yasir, S. J. A., & Youssef, S. A. (2020). Activity of
Trichoderma spp. against Erwinia carotovora causal
agent of potato tuber soft rot. Plant Archives, 20,
115–118.
Swain, H., Adak, T., Mukherjee, A. K., Sarangi, S., Samal,
P., Khandual, A., ... & Zaidi, N. W. (2021). Seed
biopriming with Trichoderma strains isolated from
tree bark improves plant growth, antioxidative defense
system in rice and enhance straw degradation capac-
ity.Frontiers in Microbiology,12, 633881.
Swaminathan, J., van Koten, C., Henderson, H. V., Jackson, T.
A., & Wilson, M. J. (2016). Formulations for delivering
Trichoderma atroviridae spores as seed coatings, effects
of temperature and relative humidity on storage stability.
Journal of Applied Microbiology, 120(2), 425–431.
Szabó, M., Csepregi, K., Gálber, M., Virányi, F., & Fekete, C.
(2012). Control plant-parasitic nematodes with Tricho-
derma species and nematode-trapping fungi: The role of
chi18-5 and chi18-12 genes in nematode egg-parasitism.
Biological Control, 63(2), 121–128.
Taha, M. A., Ismaiel, A. A., & Ahmed, R. M. (2021). 6-pentyl-
α-pyrone from Trichoderma koningii induces systemic
Eur J Plant Pathol
1 3
Vol:. (1234567890)
resistance in tobacco against tobacco mosaic virus. Euro-
pean Journal of Plant Pathology, 159(1), 81–93.
Tamandegani, P. R., Sharifnabi, B., Massah, A., & Zahravi,
M. (2021). Induced reprogramming of oxidative stress
responses in cucumber by Trichoderma asperellum (Iran
3062C) enhances defense against cucumber mosaic
virus. Biological Control, 164, 104779.
Tamizi, A. A., Mat-Amin, N., Weaver, J. A., Olumakaiye, R.
T., Akbar, M. A., Jin, S., ... &Alberti, F. (2022). Genome
sequencing and analysis of Trichoderma (Hypocreaceae)
isolates exhibiting antagonistic activity against the
papaya dieback pathogen Erwinia mallotivora. Journal
of Fungi,8(3), 246.
Tang, G. T., Li, Y., Zhou, Y., Zhu, Y. H., Zheng, X. J., Chang,
X. L., ... & Gong, G. S. (2022). Diversity of Trichoderma
species associated with soil in the Zoige alpine wetland
of Southwest China.Scientific Reports,12(1), 21709.
Tariq-Javeed, M., Farooq, T., Al-Hazmi, A. S., Hussain, M.
D., & Rehman, A. U. (2021). Role of Trichoderma as a
biocontrol agent (BCA) of phytoparasitic nematodes and
plant growth inducer. Journal of Invertebrate Pathology,
183, 107626.
Thambugala, K. M., Daranagama, D. A., Phillips, A. J., Kan-
nangara, S. D., & Promputtha, I. (2020). Fungi vs. fungi
in biocontrol: An overview of fungal antagonists applied
against fungal plant pathogens. Frontiers in cellular and
infection microbiology, 10, 604923.
Tilman, D. (1998). The Greening of the Green Revolution.
Nature, 396, 211–212.
Tilman, D. (1999). Global Environmental Impacts of Agricul-
tural Expansion: The Need for Sustainable and Efficient
Practices. Proceedings of the National Academy of Sci-
ences USA, 96, 5995–6000.
Topping, C. J., Aldrich, A., & Berny, P. (2020). Overhaul
environmental risk assessment for pesticides. Science,
367(6476), 360–363.
Tripathi, R., Keswani, C., & Tewari, R. (2021). Trichoderma
koningii enhances tolerance against thermal stress by
regulating ROS metabolism in tomato (Solanum lyco-
persicum L.) plants. Journal of Plant Interaction, 16,
116–126.
Tulasne, L. R., & Tulasne, C. (1865). Selecta Fungorum Car-
pologia, 3. Paris.
Tyśkiewicz, R., Nowak, A., Ozimek, E., & Jaroszuk-Ściseł,
J. (2022). Trichoderma: The current status of its appli-
cation in agriculture for the biocontrol of fungal phy-
topathogens and stimulation of plant growth. Interna-
tional Journal of Molecular Sciences, 23(4), 2329.
Verma, M., Brar, S., Tyagi, R., Surampalli, R., & Valero, J.
(2007). Antagonistic fungi, Trichoderma spp.: Panoply
of biological control. Biochemical Engineering Jour-
nal, 37, 1–20.
Vinale, F., Sivasithamparam, K., Ghisalberti, E. L., Marra,
R., Woo, S. L., & Lorito, M. (2008). Trichoderma–
plant–pathogen interactions. Soil Biology and Bio-
chemistry, 40(1), 1–10.
Vinale, F., Flematti, G., Sivasithamparam, K., Lorito, M.,
Marra, M., Skelton, B. W., & Ghisalberti, E. L. (2009).
Harzianic acid, an antifungal and plant growth promot-
ing metabolite from Trichoderma harzianum. Journal
of Natural Products, 72, 2032–2035.
Vitti, A., Pellegrini, E., Nali, C., Lovelli, S., Sofo, A., Vale-
rio, M., ... & Nuzzaci, M. (2016). Trichoderma har-
zianum T-22 induces systemic resistance in tomato
infected by cucumber mosaic virus.Frontiers in plant
science,7, 1520.
Waghunde, R. R., Shelake, R. M., & Sabalpara, A. N. (2016).
Trichoderma: A significant fungus for agriculture and
environment. African Journal of Agricultural Research,
11, 1952–1965.
Węgrzyn, E., & Górzyńska, K. (2019). Influence of the fungal
hyperparasite Trichoderma harzianum on the growth of
Epichloë typhina, an agent of choke disease in grasses.
Journal of Plant Diseases and Protection, 126, 39–45.
Weindling, R. (1932). Trichoderma lignorumas a parasite of
other soil fungi. Phytopathology, 22, 837–45.
Weindling, R. (1934). Studies on a lethal principle effective in
the parasitic action of Trichoderma lignorumonRhizoc-
tonia solani and other soil fungi. Phytopathology, 24,
1153–1179.
Wells, H., Bell, D., & Jaworski, A. (1972). Efficacy of Tricho-
derma harzianumas a biological control for Sclerotium
rolfsii. Phytopathology, 62, 442–447.
Woo, S. L., Ruocco, M., Vinale, F., Nigro, M., Marra, R., Lom-
bardi, N., & Lorito, M. (2014). Trichoderma-based prod-
ucts and their widespread use in agriculture. The Open
Mycology Journal, 8(1), 71–126.
Xie, X., Zhao, Z., Yang, H., Pan, H., Zhu, C., Hu, J., & Bai, Y.
(2022). Nigirpexin E., a new azaphilone derivative with
anti-tobacco mosaic virus activity from soil-derived fun-
gus Trichoderma afroharzianum LTR-2. The Journal of
Antibiotics, 75(2), 117–121.
Xiong, H., Xue, K., Qin, W., Chen, X., Wang, H., Shi, X., ..
& Wang, C. (2018). Does soil treated with conidial for-
mulations of Trichoderma spp. attract or repel subterra-
nean termites? Journal of economic entomology111(2),
808–816.
Xu, H., Yan, L., Zhang, M., Chang, X., Zhu, D., Wei, D., ... &
Yang, W. (2022). Changes in the density and composi-
tion of rhizosphere pathogenic Fusarium and beneficial
Trichoderma contributing to reduced root rot of inter-
cropped soybean.Pathogens,11(4), 478.
Yan, Y., Mao, Q., Wang, Y., Zhao, J., Fu, Y., Yang, Z., ...&
Ahammed, G. J. (2021).Trichoderma harzianum induces
resistance to root-knot nematodes by increasing second-
ary metabolite synthesis and defense-related enzyme
activity in Solanum lycopersicum L. Biological Con-
trol,158, 104609
Yan, L., & Khan, R. A. A. (2021). Biological control of bac-
terial wilt in tomato through the metabolites produced
by the biocontrol fungus, Trichoderma harzianum.
Egyptian Journal of Biological Pest Control, 31(1),
1–9.
Yang, Z., Yu, Z., Lei, L., Xia, Z., Shao, L., Zhang, K., & Li,
G. (2012). Nematicidal effect of volatiles produced by
Trichoderma sp. Journal of Asia-Pacific Entomology,
15(4), 647–650.
Yassin, M. T., Mostafa, A. A. F., Al-Askar, A. A., Sayed, S.
R., & Rady, A. M. (2021). Antagonistic activity of
Trichoderma harzianum and Trichoderma viride strains
against some fusarial pathogens causing stalk rot disease
Eur J Plant Pathol
1 3
Vol.: (0123456789)
of maize., in vitro. Journal of King Saud University-Sci-
ence, 33(3), 101363.
Yassin, M. T., Mostafa, A. A. F., & Al-Askar, A. A. (2022).
In vitro antagonistic activity of Trichoderma spp. against
fungal pathogens causing black point disease of wheat.
Journal of Taibah University for Science, 16(1), 57–65.
Yobo, K. S., Mngadi, Z. N. C., & Laing, M. D. (2019). Effi-
cacy of two potassium silicate formulations and two
Trichoderma strains on Fusarium head blight of wheat.
Proceedings of the National Academy of Sciences., India
Section B: Biological Sciences, 89(1), 185–190.
You, J., Li, G., Li, C., Zhu, L., Yang, H., Song, R., & Gu, W.
(2022). Biological control and plant growth promotion
by volatile organic compounds of Trichoderma koningi-
opsis T-51. Journal of Fungi, 8(2), 131.
Zahran, Z., Nor, N. M. I. M., Dieng, H., Satho, T., & Ab Majid,
A. H. (2017). Laboratory efficacy of mycoparasitic fungi
(Aspergillus tubingensis and Trichoderma harzianum)
against tropical bed bugs (Cimex hemipterus)(Hemiptera:
Cimicidae). Asian Pacific Journal of Tropical Biomedi-
cine, 7(4), 288–293.
Zehra, A., Aamir, M., Dubey, M. K., Ansari., W. A., Meena,
M., Swapnil P., ... & Lee, J. (2023).Enhanced protection
of tomato against Fusarium wilt through biopriming with
Trichoderma harzianum. Journal of King Saud Univer-
sity-Science,35(2), 102466
Zhang, Q., Zhang, J., Yang, L., Zhang, L., Jiang, D., Chen, W., & Li,
G. (2014). Diversity and biocontrol potential of endophytic
fungi in Brassica napus. Biological Control, 72, 98–108.
Zhang, S., Gan, Y., Ji, W., Xu, B., Hou, B., & Liu, J. (2017).
Mechanisms and characterization of Trichoderma lon-
gibrachiatum T6 in suppressing nematodes (Heterodera
avenae) in wheat. Frontiers in Plant Science, 8, 1491.
Zhang, Y. Q., Zhang, S., Sun, M. L., Su, H. N., Li, H. Y.,
Zhang, Y. Z., ... & Song, X. Y. (2022). Antibacterial
activity of peptaibols from Trichoderma longibrachia-
tum SMF2 against gram-negative Xanthomonas oryzae
pv. oryzae, the causal agent of bacterial leaf blight on
rice.Frontiers in Microbiology,13, 1034779.
Zhou, X. X., Li, J., Yang, Y. H., Zeng, Y., & Zhao, P. J. (2014).
Three new koninginins from Trichoderma neokongii
8722. Phytochemisty Letters, 8, 137–140.
Zhou, Y. M., Ju, G. L., Xiao, L., Zhang, X. F., & Du, F. Y.
(2018). Cyclodepsipeptides and sesquiterpenes from
marine-derived fungus Trichothecium roseumand their
biological functions. Marine Drugs, 16, 519.
Zhou, Y., Wang, Y., Chen, K., Wu, Y., Hu, J., Wei, Y., ... &
Denton, M. D. (2020). Near-complete genomes of two
Trichoderma species: A resource for biological control
of plant pathogens. Molecular Plant-Microbe Interac-
tions,33(8), 1036–1039.
Springer Nature or its licensor (e.g. a society or other partner)
holds exclusive rights to this article under a publishing
agreement with the author(s) or other rightsholder(s); author
self-archiving of the accepted manuscript version of this article
is solely governed by the terms of such publishing agreement
and applicable law.